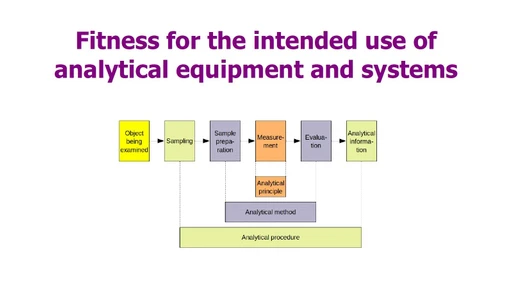
The importance of being fit for purpose
Eurachem: The importance of being fit for purpose
Introduction
Millions of tests, measurements and examinations are made every day in thousands of laboratories around the world. There are innumerable reasons underpinning them, for example: as a way of valuing goods for trade purposes; supporting healthcare and construction; checking the quality and safety of food and feed; and in forensic analysis and environmental monitoring. Virtually every aspect of society is supported in some way by analytical work.
The cost of carrying out these measurements is high and additional costs may arise from decisions made on the basis of the results. For example, tests showing food to be unfit for consumption may result in compensation claims. In addition, tests confirming the presence of banned drugs could result in fines or imprisonment. Clearly it is important to make a correct measurement and be able to show that the result is correct.
The validation/verification process
Most analysts know that method validation/verification is important, but exactly why, how and when it should be done is not always clear.
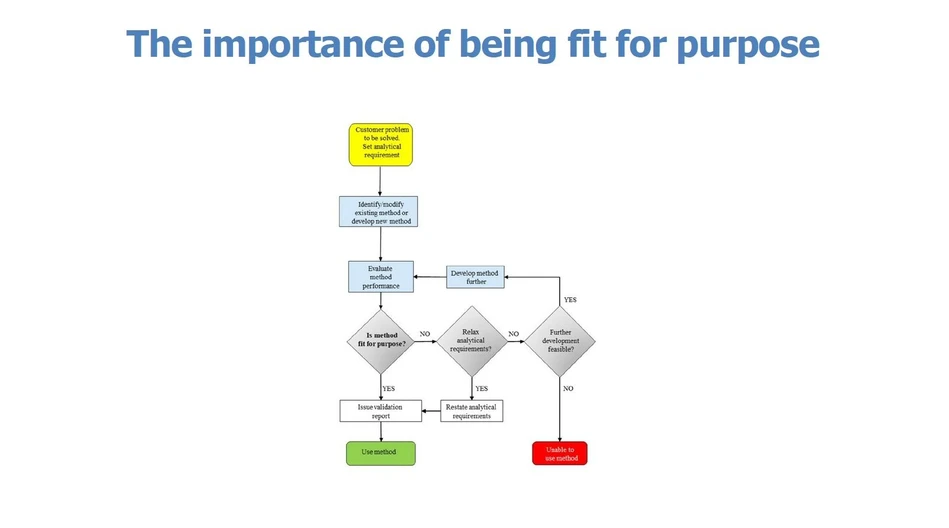
Method validation/verification is the process whereby the laboratory demonstrates whether or not a method is ‘fit for purpose’ (Fig. 1). This means that the tests carried out should be appropriate with respect to uncertainty, cost, time etc. The final report should present analytical data in such a way that the customer can, readily, interpret it and draw appropriate conclusions.
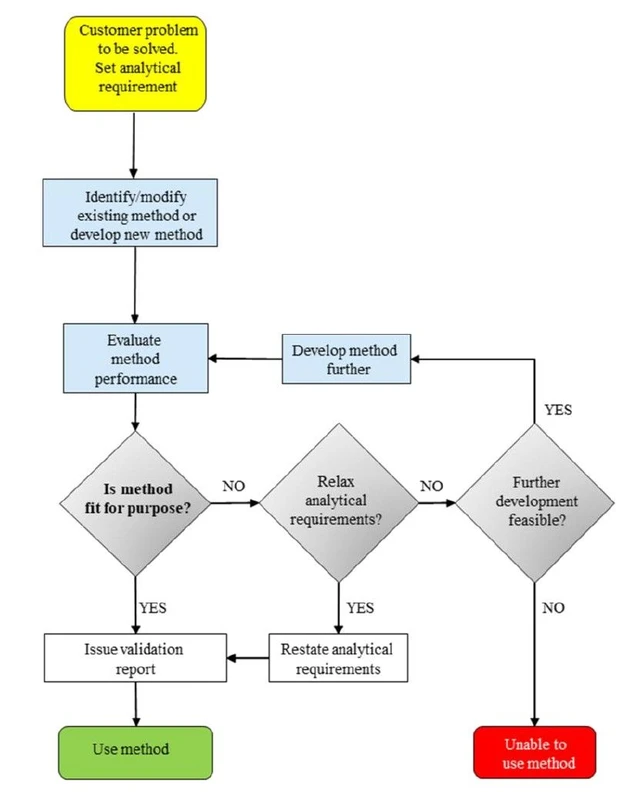 Eurachem: Fig. 1. The method validation/verification process. The laboratory ‘translates’ the customer’s problem into an analytical requirement, i.e. the method performance required to solve the problem. Method validation/verification includes a stage where various performance characteristics are evaluated and then compared with analytical requirements.
Eurachem: Fig. 1. The method validation/verification process. The laboratory ‘translates’ the customer’s problem into an analytical requirement, i.e. the method performance required to solve the problem. Method validation/verification includes a stage where various performance characteristics are evaluated and then compared with analytical requirements.
The Eurachem Guide
A Guide from Eurachem (Fig. 2) gives practical advice on how method validation/verification can be accomplished. The Guide and its supplements [1-4]:
- Introduce the basic concepts related to method validation and method verification;
- Indicate how to plan, record and report validation and verification studies to best support the statement of ‘fitness for purpose’;
- Provide key definitions and the rationale behind the experiments for assessing the various performance characteristics and related topics (Fig. 3);
- Include quick reference tables that suggest experiments together with the necessary statistical calculations for evaluation and reporting each performance characteristic;
- Provide support to the analyst on how to make the best use of method validation data for setting up an internal quality control plan.
 Eurachem: Fig. 2. The Eurachem Guide is available free of charge at www.eurachem.org.
Eurachem: Fig. 2. The Eurachem Guide is available free of charge at www.eurachem.org.
How should methods be validated/verified?
The guide focuses on a single-laboratory approach to method validation/verification and gives guidance on planning, carrying out and reporting method validation and verification studies. Any method validation or verification study will require the laboratory to investigate several performance characteristics:
- Precision
- Repeatability
- Intermediate precision
- Trueness
- Selectivity
- Working range
- Analytical sensitivity
- Limit of detection
- Limit of quantification
- Ruggedness
Exactly which characteristics are studied will depend on the analytical application. Verification of the performance of a standard method requires substantially less work than validation of a method developed in-house. Legislative/sectoral requirements must also be considered.
Sampling and subsampling can be part of the measurement or testing procedure and must, in those cases, be validated.
- H. Cantwell (ed.) Eurachem Guide: The Fitness for Purpose of Analytical Methods – A Laboratory Guide to Method Validation and Related Topics, (3rd ed. 2025). Available from www.eurachem.org.
- V. Barwick (ed.), Planning and Reporting Method Validation Studies – Supplement to Eurachem Guide on the Fitness for Purpose of Analytical Methods (2 nd ed. 2025). Available from www.eurachem.org.
- H. Cantwell (ed.) Blanks in Method Validation – Supplement to Eurachem Guide on The Fitness for Purpose of Analytical Methods, (2 nd ed. 2025). Available from www.eurachem.org.
- M. H. Ramsey, P. D. Rostron, F. C. Raposo (eds.) Eurachem/EUROLAB/CITAC/Nordtest/AMC Guide: Validation of Measurement Procedures that Include Sampling, Eurachem (1st ed. 2024). Available from www.eurachem.org.