New trends in liquid chromatography and their utilization in analysis of beer and brewery raw materials. Part 2. Determination of cis/trans- isomers -iso-α-acids in beer using Ultra Performance Liquid Chromatography

Pixabay/carolineandrade: New trends in liquid chromatography and their utilization in analysis of beer and brewery raw materials. Part 2. Determination of cis/trans- isomers -iso-α-acids in beer using Ultra Performance Liquid Chromatography
The new UHPLC method was compared with the HPLC method, which is used for iso-α-acid determinations in RIBM. The comparison was performed with respect to the speed of analysis, separation efficiency, mobile phase consumption, and finally repeatability. The results of this study determined all the expected advantages of the UHPLC; while maintaining the resolution this method reduced analysis time 2.5 times compared to the previous HPLC. In addition, the UHPLC reduces the consumption of organic solvents 10 times. Apart from the economic benefits the UHPLC provides a high precision of the measurement, which was verified as repeatability of the UHPLC for both forms of iso-α-aids. Especially the trans-iso-α-isomers are significant indicators of beer aging during storage and transport. It was found that the repeatability of UHPLC for the trans-isomers is better than the repeatability of HPLC.
1 INTRODUCTION
Iso-α-acids are formed during the brewing process in two steric forms, cis- and trans-, in a ratio of 2:1 in favor of the more stable cis-form. The factors influencing the beer aging process cause a faster decrease of the trans-form, which results in both a decrease of beer bitterness and the formation of sensory undesirable compounds. Therefore modern technology processes, which look for the ways of slowing down beer aging, focus on the possibility how to reduce the drop in the concentration of iso-α-acids (De Cooman et al., 2000).
During the development of analytical methods for determination of cis-/trans-iso-α-acids, close attention is paid to the isolation of these compounds from beer (SPE extraction) and to the complete separation and sufficiently sensitive determination in the beer using the liquid chromatography methods. In the present, classical HPLC methods are used for the purpose. However, new chromatography techniques based on separation on columns packed with sub-2 μm particles are being increasingly used in the analysis of beer. The efficiency of these columns does not change when the linear velocity increases. The chromatography employing these columns is called Ultra High Performance Chromatography (UHPLC). The principles of this method were described in issue 2, 2012 of the journal. An identical and often better separation can be achieved using UHPLC in comparison with HPLC; furthermore, the analysis time is significantly shorter. The higher backpressure, which is caused by extremely small particle size in UHPLC columns, has been overcome using special and newly developed pumps, absolute narrowness of the whole system, and a number of technical elements forming the new UHPLC system. Apart from the separation speed a further advantage of UHPLC is very low consumption of organic solvents (up to 10-fold lower compared to HPLC), and a very small injection volume (1–2 μl).
The UHPLC was used for the development of a new method of cis- and trans-iso-α-acids determination in beer. The separation parameters were compared with the classical HPLC method, which is used in RIBM (Research Institute of Brewing and Malting). Besides comparing the speed of analysis and separation efficiency the values of repeatability (r₉₅) of the two methods were confronted.
The separation of iso-α-acids and their isomers in beer is usually performed on the reversed C18 stationary phase. The mobile phase is prepared as a buffer with acid pH or the aqueous solution of any acid. Acetonitrile (ACN) is usually used as organic modifier. The development of highly resistant stationary phases in a wide pH range reaching up to alkali values gives rise to new methods of separation using this pH range (Hofta et al., 2007). The good selectivity of these columns enables the simultaneous determination of iso-α-acids, their oxidative products, and different forms of hydrogenated iso-α-acids. The HPLC method of determination of cis- and trans- isomers of iso- α-acids under neutral or weak alkali conditions (Harms et Nitzsche, 2001) was used in the study concerning the cis-/trans- ratio changes during beer storage under various temperature conditions. (Straková et al., 2007). However, it is necessary to note that alkali pH causes the transformation of α-acids to iso-α-acids and thus a shift in the equilibrium (Basařová et al., 2010). The dependence of isomerization on the analysis at alkaline pH has not yet been documented.
The original variant of mobile phase with acidic pH was chosen for the purpose of our study.
2 MATERIAL AND METHODS
Domestic beers with the bitterness ranging from 25 to 34 mg/l were chosen for comparing the efficiency of UHPLC and HPLC methods in the determination of cis- and trans-iso-α-acids. The iso-α-acids were isolated from the beer using the SPE technique on the extraction columns Phenomenex according to a previously described procedure (Jurkova et al., 2003).
The iso-α-acid dicyclohexylamine containing 62.3% of iso-α-acids and highlighted as DCHA-Iso, ICS-13, which is an international calibration standard (ICS), was used as an external standard for the calibration in both methods.
The stock solution of the calibration standard was prepared by dilution of ICS-13 (20 mg) weighed with the precision of 0.1 mg in 100 ml of gradient grade methanol (99.8%, Merck). Prior to the dilution, the methanol was acidified with phosphoric acid (p.a. Merck) in an acid-methanol ratio of 0.5 : 1000 (v/v). For HPLC and UHPLC, the calibration standard was diluted tenfold and twentyfold with acidified methanol, respectively.
HPLC chromatographic conditions
The separation was performed on a reversed phase column (Alti-ma C18, 5 μm, 150 x 4.6 mm, Alltech) protected by a C18 pre-column (4 x 3 mm, Phenomenex). The linear gradient was applied and two-component mobile phase (A) and (B) was used. Phase (A) with pH 2.7 was prepared from ultrapure water (Millipore) with a maximum organic compound content of 5 ppb, and phosphoric acid. Phase (B) was gradient grade acetonitrile (≥99.9%, Sigma Aldrich). The linear gradient started with 52 % of ACN, the content of ACN linearly in-creasing within 30 min to the final concentration of 67 %. The equilibration time was 10 minutes. The flow rate was 1.5 ml/min and the column temperature was 40 °C. The analytes were detected in the UV area at 275 nm. The injection volume was 10 μl.
The Spectra SYSTEM (TSP, USA) liquid chromatograph with PDA detector was used for the analysis. The data collection and data processing was carried out with the ChromQuest software for Windows NT.
UHPLC chromatographic conditions
UHPLC analyses were performed on the BEH C18 column (2.1 x 50 mm I.D., 1.7 μm), Waters, the column temperature was 40 °C and the mobile phase flow was 0.4 ml/min. The diode array detector (PDA) was used for detection; analytes were detected at 275 nm. The two-component mobile phase was composed of acetonitrile (B) and aqueous solution of acids or buffers (ultrapure wa-ter, Millipore) with pH ranging from 1.6 to 3.08 (A). During the optimization the following mobile phase (A) were tested:
TFA, pH 1.6 (0.1% vol., prepared as a solution of 100 μl TFA, 99.95% ULC/MS, Biosolve, Netherlands) in 100 ml of ultrapure water;
TFA, pH 2.7 (0.01% TFA vol., prepared as a solution of 10 μl, 99.95% ULC/MS, Biosolve, Netherlands) in 100 ml of ultrapure water; using of this conditions was achieved the best separation, thus,
this phase was finally chosen as optimal (see section “3. Results and Discussion”);
Ammonium formate, 5 mM, pH 3.08 (prepared using titration of 5 mM formic acid (99%, Merck, Germany) with aqueous solution of ammonium hydroxide, 29%, A.C.S. reagent, Sigma Aldrich, Germany to the final pH of 3.08).
The gradient elution was optimized in terms of the minimal analysis time and maximal resolution of determined compounds. The final gradient conditions were as follows: the gradient started at 48% of phase (B) and grew linearly up to 55% (B) within 11 min. The subsequent isocratic step at 55% (B) and equilibration step starting at final condition of 48% (B) were 2 min and 3 min, respectively. The injection volume was 2 μl.
UHPLC analyses were performed on Acquity UPLCTM (Waters) chromatograph with the 2996 PDA detector operating in the range from 194 to 600 nm. Data were processed with Empower 2 software (Waters). Chromatograms were extracted at 275 nm before interpretation, data sample rate was set at 20 pts/s and filter constant was 0.5.
3 RESULTS AND DISCUSION
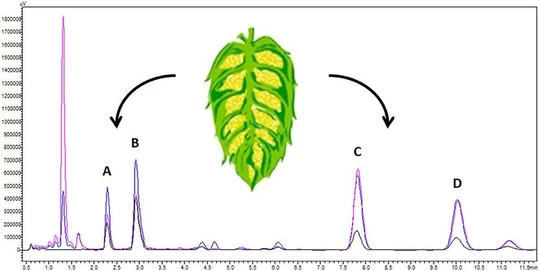
Comparison of UHPLC and HPLC chromatograms of separated iso-α-acids isomers is given in Fig. 1 and Fig. 2, respectively. Both chromatograms were measured under optimal UHPLC and HPLC conditions described in Materials and Methods. Fig. 1 confirms that the UHPLC method is faster than HPLC; the analysis time was reduced 2.5 times in comparison with HPLC method. In the UHPLC, the last eluting peak of cis-iso-ad-humulone had the retention time of 4.9 min, the total analysis time including the wash step was 13 min, and equilibration step was 3 min. Thus, the total analysis time was 16 min. In the HPLC, the last eluting peak of cis-iso-ad-humulone eluted at 12 min, the gradient wash step lasted 30 min, and the equilibration step was 10 min, the total HPLC analysis time being thus 40 min. In both events, the gradient is continuing also after elution of cis-iso-ad-humulone because residual α- and β-acids can elute even later. If the detection of α- and β-acids is not required (reduced method version) then it is possible to reduce the gradient by implementing a fast wash step with a high content of organic modifier after 5th and 12th min in UHPLC and HPLC mode, respectively (see Fig. 1 and 2). If the wash step is not included or if it is not sufficient, the residual α- and β-acids can accumulate on the column and can subsequently interfere in the following analyses. The comparison revealed a noticeable difference between the two methods. Apart from the marked difference in the time of analysis a huge difference in the consumption of organic solvents is evident. While the ACN consumption under our experimental conditions in the UHPLC mode is 32 ml per 10 samples, in the HPLC mode it is 360 ml per 10 samples, i.e. ten times more.
 Fig. 1 UHPLC chromatogram of real beer sample
Fig. 1 UHPLC chromatogram of real beer sample
 Fig. 2 HPLC chromatogram of real beer sample
Fig. 2 HPLC chromatogram of real beer sample
The transformation of the HPLC method to the UHPLC and its optimization revealed a strong dependence of the retention time of studied compounds, iso-α-acids, on the mobile phase pH. Even small changes in the monitored pH range (from 1.6 to 3.08) give rise to a significant shift of the retention under the maintenance of the organic modifier gradient. A very good separation was achieved in UHPLC mode at pH 2.7, when the retention time of the last eluting peak belonging to iso-α-acid (cis-iso-ad-humulone) was 4.9 min. This pH proved as optimal even for usage in HPLC mode, where the retention time of this peak is 12 min. When the pH was decreased to 1.6 (0.1% TFA) the retention times was extended four times and the last determined peak of cis-iso-adhumulone eluted at 19th min. Additionally, the peaks of trans-iso-ad-humulone and cis-iso-n-humulone were not separated. On the contrary, when the pH was increased to 3.08 (5 mM ammonium formate) the retention times were notice-ably reduced. All iso-α-isomers eluted in the range from 1 to 2 min, but isomeric forms cis- and trans- of any acids were not separated.
TFA, formic acid, and phosphoric acid were tested as potential additives to the aqueous part of the mobile phase. Phosphoric acid is commonly used in the original HPLC method. However, this acid was found to be unsuitable in the UHPLC mode, because all the peaks were considerably deformed. Finally, TFA proved to be the optimal additive because its usage showed the best separation parameters. During the gradient optimization it was necessary to decrease the initial content of organic modifier to 48%, because the use of the same percentage of ACN as in HPLC (52 %) caused fast peak elution with insufficient separation. This observation is in correlation with the UHPLC theory according to which the analytes pass through the column under much higher pressure (up to five times) compared to HPLC. The final content of ACN in the gradient was also changed, the percentage of 55 % being sufficient for good and fast separation instead of the original 67 %. Finally, the time of duration of the linear gradient was also reduced, specifically from 30 to 11 min.
The last step was the optimization of the injection volume. Regard-ing the different column dimension of 150 x 4.6 mm and 50 x 2.1 mm in HPLC and UHPLC, respectively, the injection volume should be reduced during the method transfer. Injection volume higher than 2 μl caused overloading of UHPLC column, which appeared in the peak splitting. When 2 μl of the sample was loaded on the column it ensured a very good separation of all determined compounds with convenient sensitivity which was in accordance with the quantification limits of the method.
The repeatability of the new UHPLC method was compared with the original HPLC method and was calculated from the values of the iso-α-concentration values in 4 real beer samples, every sample was measured in duplicate. The evaluation was carried out from the peak areas of both trans- and cis- forms of iso-co-humulone, iso-ad-humulone, and iso-n-humulone. The results of individual concentrations and repeatability values of all studied isomers in both UHPLC and HPLC systems are given in Tab. 1 and 2. The repeatability r₉₅ was calculated according to the formula r₉₅ = sᵣ x 2,8, where sr is standard deviation calculated for various, but similar, samples measured un-der the conditions of repeatability:
sᵣ² = ΣDᵢ²/2n, where Dᵢ is the range of values from two determinations.
 Tab. 1 Determination of cis- and trans- forms of iso-α-acids using the HPLC method
Tab. 1 Determination of cis- and trans- forms of iso-α-acids using the HPLC method
 Tab. 2 Determination of cis- and trans- forms of iso-α-acids using the UHPLC method
Tab. 2 Determination of cis- and trans- forms of iso-α-acids using the UHPLC method
The UHPLC method affords a comparable or even better repeat-ability compared to the HPLC for the all cis- and trans-iso-α-acids with the exception of cis-isomers of co-humulone and n-humulone. This could be probably caused by better geometry of UHPLC peaks leading to better peak integration and thus, better quantification.
Furthermore, it could be established that the use of the UHPLC method results in a smaller variance and thus a more accurate con-centration determination of trans- forms, which are considered as an indicator of beer stability or beer ageing during storage or transport.
The Tab. 3 summarizes described differences and advantages of newly developed UHPLC method comparing the original HPLC method.
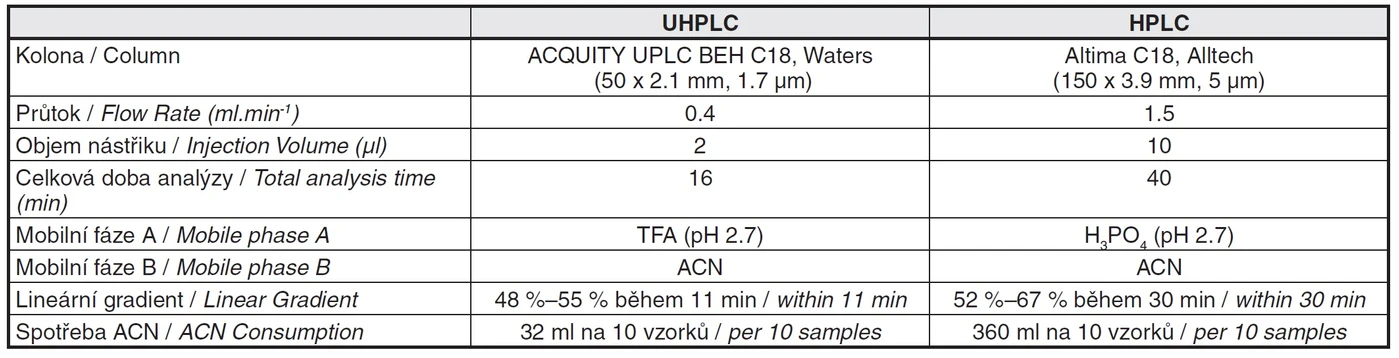 Tab. 3 Comparison of UPLC and HPLC method parameters
Tab. 3 Comparison of UPLC and HPLC method parameters
4 CONCLUSION
The new generation of UHPLC liquid chromatography based on more advanced instrumental technique is not only more economical (reduced analysis time with low consumption of solvents) but it also shifts the analysis of beer to the area with more accurate measurement and lower variance of results, which was demonstrated in this study on the example of determination of steric iso-α-acid isomers.