New Trends in Liquid Chromatography and Their Utilization in Analysis of Beer and Brewery Raw Materials. Part 1. Theoretical Introduction

Pixabay/nurfayozagzamov1004: New Trends in Liquid Chromatography and Their Utilization in Analysis of Beer and Brewery Raw Materials. Part 1. Theoretical Introduction
In connection with increasing requirements on high throughput analyses in the laboratories the instrumental techniques undergo continuous improvement. This article is focused on the progress in the most widespread separation technique, the liquid chromatography. Its novel modification, Ultra High Performance Liquid Chromatography (UHPLC), uses the principle of separation on porous “sub-2 μm“ particles. The advantages of UHPLC over the traditional HPLC are a nine-fold reduction of analysis time, double improvement of resolution, and triple improvement of sensitivity. It results not only in time saving but also in the cost of energy and organic solvents and their ecological disposal. An alternative to UHPLC is the use of columns with core-shell particles and/or monolithic columns. The UHPLC has already been successfully applied in the analysis of beer and brewery materials.
1 INTRODUCTION
High performance liquid chromatography represents nowadays the most widespread separation technique applied commonly in pharmacy, toxicology, clinical analysis as well as in various research fields (Stroh et al. 2008, Pratet et al. 2004, Klein et Rivera 2004) and, last but not least, in food chemistry, namely for the analysis of beer and its raw materials (e.g. Čulík et al. 2009).
Increasing requirements on high throughput of samples motivate a continuous improvement of analytical techniques. During the last decade the HPLC technique has also become “faster”. There are two basic approaches to “fast chromatography”. The first one leads to the development of a completely new generation of chromatographic columns and instrumentation operating at 100 MPa (15 000 PSI) (Swartz 2005). This method was established and termed as Ultra High Performance Liquid Chromatography (UHPLC). The second approach maintains the classical HPLC instrumentation that works at a maximal pressure of 40 MPa (6000 PSI). However, only new types of HPLC instruments comply with this pressure limitation. Analysis acceleration is achieved by the use of special columns, namely monolithic columns and columns with core-shell particles.
2 UHPLC –ULTRA HIGH PERFORMANCE LIQUID CHROMATOGRAPHY
The complex process of chromatographic separation is determined by a set of analyte interactions between mobile (solvent) and stationary phase (in simplified terms column packing). During analyte passage through the chromatographic column the analyte is carried by the mobile phase. The analyte has specific affinity to the mobile phase (it is soluble in it) and simultaneously interacts with the stationary phase (mass transfer). The contact of analyte with the stationary phase is mediated by “diffusion”. There exist three types of diffusion during the chromatographic process - longitudinal diffusion (a movement of the analyte in the mobile phase), turbulent diffusion (a movement of the analyte to the surface of the stationary phase particles and back), and diffusion inside the pores of stationary phase particles in various directions (mass transfer of analyte to the stationary phase and back). The mathematic formulation of the contribution of individual diffusion modes is called van Deemter equation
H = a(dp) + b/u + c(dp)²u
The equation determines the relationship between the height of theoretical plate H (inverse value of column efficiency) and linear velocity u of the mobile phase. The contribution a (longitudinal diffusion) is influenced only by particle size, contribution b (turbulent diffusion) is inversely proportional to the linear velocity u, and contribution c (mass transfer) is directly proportional to a square of particle size of the stationary phase.
 Fig. 1 Van Deemter curves for 10, 5, 3, and 1.7 μm particle size
Fig. 1 Van Deemter curves for 10, 5, 3, and 1.7 μm particle size
Since the seventies, when the particles with 10 μm diameter were used, a huge progress has been achieved in the development of technologies for generation of smaller particles, which led to permanent improvement of separation parameters. Fig. 1 shows four van Deemter curves for different size particles that are commonly used (10, 5 a 3 μm and newly 1.7 μm). It follows from the figure that decrease of particle size leads to increase of separation efficiency (in other words to a decrease in plate height). This reflects the fact that the use of the smaller particle size ensures better mass transfer and simultaneously minimizes band broadening. It has impact on increased efficiency and, in addition, makes it possible to work with higher mobile phase flow and achieve thus a faster separation. This consideration led to the development of “sub-2 μm particles” (Mellors et Jorgenson 2004) providing an impetus for evolving Ultra-High Performance Liquid Chromatography (UHPLC). Using this technology, the analysis time can be reduced 9 times, sensitivity can be improved 3 times, and resolution 2 times. Such reduction of the time of analysis results not only in saving of energy and manhours but also in 10-fold reduced demand for organic solvents and in reduced costs of safe waste disposal. Evaluation of analytic parameters and comparison of general HPLC and UHPLC chromatograms is demonstrated in Fig. 2. It should be noted that columns packed with sorbent with such small particle size generate much higher back pressure than common chromatographic columns. Hence, a new UHPLC instrument had to be developed to be able to work under ultra-high pressures such as 100 Mpa (15 000 PSI).
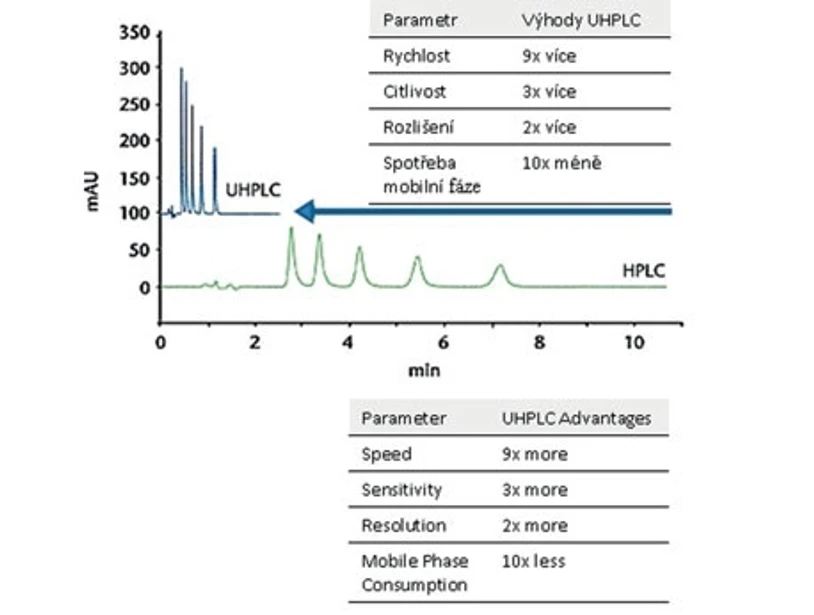 Fig. 2 Comparison of UHPLC and HPLC chromatograms (The table lists the UHPLC characteristics superior to HPLC)
Fig. 2 Comparison of UHPLC and HPLC chromatograms (The table lists the UHPLC characteristics superior to HPLC)
Fig. 3 demonstrates the size of new 1.7 μm particle in comparison with both a common 5 μm particle and the diameter of human hair (60 μm).
 Fig. 3 Size comparison of 1.7 μm hybrid particle (C), common 5 μm particle (A) and diameter of a human hair 60 μm (B)
Fig. 3 Size comparison of 1.7 μm hybrid particle (C), common 5 μm particle (A) and diameter of a human hair 60 μm (B)
The information about the first commercially available Ultra High Performance Liquid Chromatography (UHPLC) (Schwartz 2005) system including subsequently the principle of novel ultra-fast chromatography, description of new sub-2 μm particles and related new column chemistry and technology (Mellors 2004), full-scale hardware, new designed UV detector, and connection with MS detectors (Plumb et al. 2004) was published in 2004. These columns, Acquity UPLC BEH C18 (Waters), packed with 1.7 μm particles are based on BEH (bridged ethylene hybrid) technology (Swartz et Murphy 2005). Between silanol groups are inserted hybrid ethylene bridges (see Fig. 4), which function as end-capping groups. Furthermore, the bridges fix the overall structure of this silica gelbased phase. This endows the columns with excellent mechanical robustness (max. pressure limit 15000 PSI) and chemical stability in a wide pH range. These columns can therefore be used in alkali pH range up to pH 12; acidic range up to pH 1-2, but this value depends on the column ligand used. In addition, these columns are resistant to “column bleeding”. A wide range of columns with various ligands is currently available on the market. They include BEH C18 and C8 (linear alkyl chain for classical reverse chromatography), BEH Shield RP18 (alkyl chain with inserted polar carbamate group, which combines hydrophobic and hydrophilic properties of the column and is convenient for polyphenolic compounds), BEH Phenyl (phenyl ligand on C6 alkyl chain convenient for the compounds with benzene nucleus), BEH Amide column (amidic group that retains well compounds too polar for reverse chromatography), BEH HILIC (BEH particles without a ligand, convenient for extremely polar compounds; elution uses aqueous phases with low organic content), etc. The lengths of these columns are 2, 3, 5, 10, and 15 cm, the most commonly used column has 5 cm. The inner diameter of the columns is mostly 2.1 mm and 1 mm when connected with UV and MS detector, respectively. Like the classical column, the column is protected by a pre-column and additionally by a special filter, which protects the column from potential impurities from the mobile phase. The small particle size poses demands on both the mobile phase and sample preparation. Mobile phase must be filtered through 0.2 μm membrane filter and the sample has to be filtered or centrifuged prior to analysis.
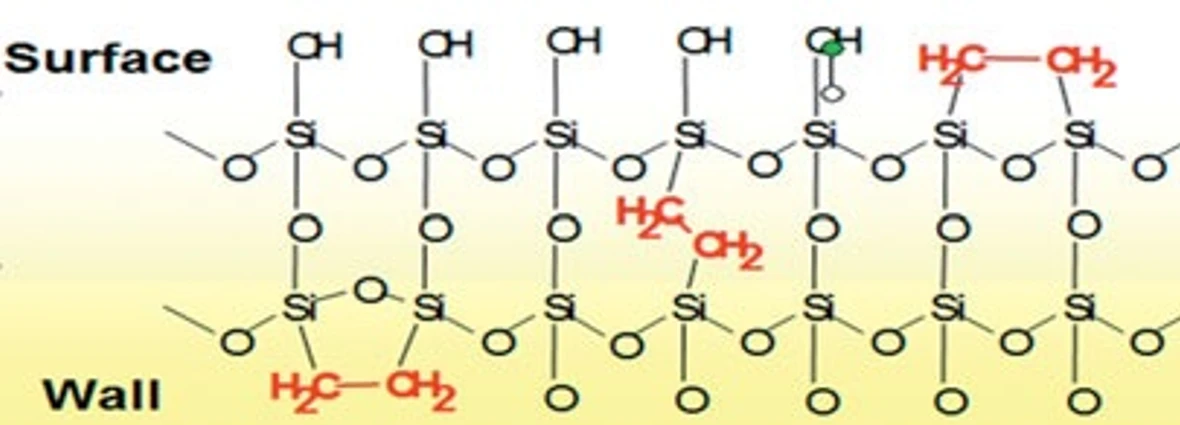 Fig. 4 Illustration of the chemical structure of the hybrid BEH particle with hybrid ethylene bridges
Fig. 4 Illustration of the chemical structure of the hybrid BEH particle with hybrid ethylene bridges
An increasing number of UHPLC columns with particle size ranging from 1.5–2 μm (e.g. Waters – Acquity BEH columns, Restek – Pinnacle® DB columns, Grace – Vision HT columns, Thermo Scientific – Hypersil GOLD, Agilent – Eclipse Plus C18 a StableBond SB-C18, Perkin Elmer – Brownlee Analytical DB C18), and the rising number of UHPLC instrument producers give evidence that UHPLC was accepted by analytical public. Apart from the Waters company (Acquity), which was the pioneer of this technology, other producers of UHPLC chromatographs with similar parameters are on the market, e.g. Agilent (1200RRLC, Infinity), ThermoElectron (Acella High Speed), Shimadzu (ProminenceUFLC), Dionex (RSLC UltiMAte 3000), Perkin Elmer (Flexar). Two synonymous terms, UPLC and UHPLC, can be found in the literature. Since UPLCTM (Ultra Performance Liquid Chromatography) technology was applied as the first in the world by the Waters Company, the term UPLC can be taken as a trade mark. Hence, UHPLC was accepted as the common term for this technology.
The advantages of UHPLC have been made use of also in the analysis of brewing materials and beer. Many applications were developed by the research team from RIBM (Research Institute of Brewing and Malting). A recent application, which was published in an international scientific journal in 2011, is the determination of ochratoxin A in brewing raw materials and beer (Běláková et al. 2011) and also in wine. The transformation of the original HPLC method to the new UHPLC method brought a reduction of the analysis time from the original 10 min to 2 min. The same result was obtained for the method of ferulic acid determination in barley and malt, in which the original analysis time of 12 min was reduced to 2 min (Běláková et al. 2010). The same authors developed more UHPC methods, which are used in RIBM in many applications. These include, apart from the determination of ferulic acid, also methods for the determination of other phenolic acids (cumaric acid etc.), vitamin E and vitamins from the B group in beer and brewery raw materials, patulin in beer, brewery raw materials, wine and nonalcoholic beverages, and/or determination of silymarine in an extract of Silybum Marianum.
In the following issues of this journal we shall present a more detailed comparison of HPLC and UHPLC method illustrated on examples of the determination of α- a β- acids in hop and hop products and determination of iso-α-acids in beer developed in RIBM.
3 CORE-SHELL PARTICLE COLUMNS
Even if the user cannot purchase a new UHPLC instrument and is bound to use a current HPLC instrument, he can still speed up the separation process. He can use columns with “fused-core silica” or synonymous “core-shell particles” working at common pressures (max 6000 PSI) that are compatible with the majority of HPLC instruments. The particles (see Fig. 5) are composed of a solid core (about 1.7 μm) surrounded by a thin porous silica layer (about 0.5 μm); their exact dimensions vary depending on the producer. The analyte can diffuse only into the pores of the thin porous ± 0.1 μm layer, where mass transfer takes place. The diameter of these 2.7 μm particles results in the generation of lower back pressure than in the case of “sub-2 particles”. At the same time, the thickness of the porous silica layer is comparable with the diameter of “sub-2 particles”. These characteristics of the “core-shell particles” ensure separation efficiency and resolution similar to UHPLC. Lastly, the analyte does not have to diffuse to the rigid core through the whole particle, which results in a shortening of diffusion path, the delay in the column, and in reduction of analysis time without deterioration of separation parameters.
Many papers published since 2010 have confirmed both the broad utility of core-shell columns in practice and their separation properties comparable with those of “sub-2” particles (Abrahim et al. 2010, Tylová et al. 2011). These columns, having a wide spectrum of various ligands, e.g. Halo (Mac-Mod Analytical), Ascentis Express (Sigma-Aldrich), Kinetex (Phenomenex) and/or Poroshell (Agilent) are currently available.
Determination of quinolones in eggs is an example of application of core-shell columns in food analysis. The column Kinetex was used and analysis time was reduced by half (Jiménez et al. 2011). The same column was also used for the first time for beer analysis for determination of iso-α-acids and some reducing forms of tetrahydroiso-α-acids (Koerner et al. 2011).
4 MONOLITHIC COLUMNS
Another approach to accelerating chromatographic process is the use of monolithic columns, which are gaining increasing popularity. In contrast to conventional stationary phases, which are formed by individual sorbent particles of defined size, the monolithic columns are formed by a single piece of porous material completely filling the interior of the column. Thus, monolithic columns do not contain any space between particles through which the majority of the flow proceeds in conventional columns. That’s why the mobile phase has to pass through the monolith pores that are much bigger than the pores in columns with common particles. Monolithic columns have two types of pores. Macropores facilitate fast convective flow of mobile phase through the monolith and significantly accelerate mass transfer between mobile and stationary phase. Mesopores provide sufficiently large surface and also a high separation capacity. This structure allows monoliths to operate at considerably high flow of mobile phase without excessive pressure increase (McCalley 2002) and without losses of separation efficiency even for separated macromolecules (peptides, synthetic polymers) (Švec 2009).
Monolithic columns are synthetized by hydrolytic polymeration of tetramethoxysilane or tetraethoxysilane in aqueous solution of acetic acid in the presence of polyethylene glycol. Manufacturing technology enables monolith preparation with accurately defined structure and with macropores and mesopores with convenient and adjustable sizes. The monolithic stationary phase was prepared, described and applied by, e.g., Prof. Tanaka’s group (Minakuchi et al. 1996). Many of these columns are already commercially available, e.g. Chromolith (Merck) or Onyx™ (Phenomenex in the licence of Merck).
As follows from the real applications and comparative studies of monolithic and conventional reversed phase columns, these columns have also found application in the area of fast chromatography and the number of their users continually increases (Samanidou et al. 2004, Nováková et al. 2004). The technology starts to be used also in food analysis. Tzanavaras et Themelis (2007) published a method of simultaneous determination of caffeine, theobromine and theophylline in coffee and non-alcoholic beverages. The analysis time did not exceed 1 min and the retention time of the last eluting caffeine was 0.7 min.
5 CONCLUSION
The huge quantity of published articles gives evidence that in the last decade UHPLC has become a favorite and widespread separation technique. It follows from these article that UHPLC is used for the separation of both small and large organic compounds such as proteins and peptides. The published data confirm the presumed advantages of UHPLC such as its high speed, sensitivity and good resolution. UHPLC technique found its use also in food analysis and begins to spread into the area of brewing and malting.




