New blood biomarkers reveal how faithfully diabetes patients take metformin
Clin Pharmacol Ther, 119: 1057-1069: Graphical abstract
Metformin is currently the first-line oral drug of choice for the treatment of type 2 diabetes. It reduces glucose production in the liver, improves tissue sensitivity to insulin, and does not cause weight gain or hypoglycemia, which sets it apart from some other antidiabetic drugs. Moreover, its long-term beneficial effects on the risk of cardiovascular complications and mortality in people with type 2 diabetes have been well established. Researchers from the Institute of Physiology CAS (IPHYS) have identified a new group of blood metabolites that could help physicians objectively monitor whether patients with type 2 diabetes are taking their metformin as prescribed.
New biomarkers for metformin use
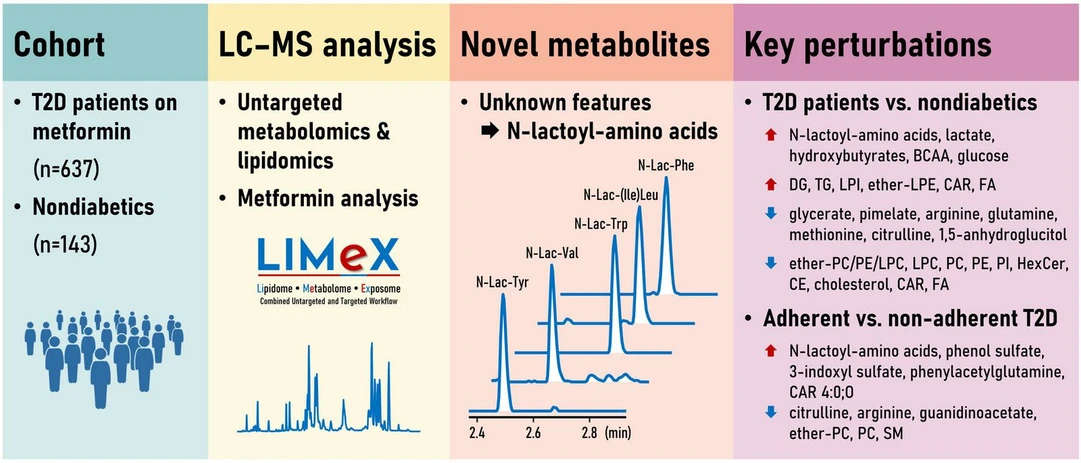
A team led by Tomáš Čajka from the Laboratory of Translational Metabolism at IPHYS analyzed plasma samples from 637 patients with type 2 diabetes treated with metformin and 143 non-diabetic controls using advanced metabolomics and lipidomics. Among 614 detected metabolites, they identified five from the group of N-lactoyl-amino acids (e.g. N‑lactoyl-phenylalanine), which had previously been seen as unknown signals due to missing reference spectra in databases. The levels of these metabolites in the blood closely reflected the plasma concentrations of metformin. In patients with the highest metformin exposure, the concentrations of these metabolites were up to 7.2 times higher than in controls.
A sensitive “fingerprint” of the drug’s effect
N-Lactoyl-amino acids are formed in cells by combining lactate and amino acids, thus reflecting the metformin-induced changes in cellular energy metabolism. “Importantly, even though they occur in plasma only at nanomolar concentrations, they reliably distinguish patients who take metformin regularly from those who take it irregularly or not at all,” emphasizes T. Čajka.
Metabolomics in practice and international collaboration
The study also provides a detailed description of broader metabolic changes in type 2 diabetes, including increases in lactate, organic acids, and branched-chain amino acids, as well as extensive remodelling of the lipid profile. In addition to T. Čajka’s team, collaborators from the Institute for Clinical and Experimental Medicine in Prague and the West Coast Metabolomics Center (University of California, Davis) also participated in the study.
The results demonstrate that advanced metabolomics approaches developed at IPHYS within the CarDia project can find highly practical applications both in clinical studies and in personalized monitoring of treatment in patients with diabetes.
The original article
Untargeted Metabolomics Identifies N-Lactoyl-Amino Acids as Dose-Responsive Plasma Biomarkers of Metformin Adherence in Type 2 Diabetes
Tomas Cajka, Jiri Hricko, Lucie Rudl Kulhava, Michaela Paucova, Michaela Novakova, Veronika Hola, Stanislava Rakusanova, Oliver Fiehn, Vojtech Skop, Ivana Lankova, Iva Miskova, Terezie Pelikanova, Martin Haluzik
Clin Pharmacol Ther, 119: 1057-1069
https://doi.org/10.1002/cpt.70205
licensed under CC-BY 4.0
Abstract
Metformin is the most widely prescribed antidiabetic drug, yet adherence remains difficult to objectively assess. Using untargeted metabolomics and lipidomics, we analyzed plasma from 637 patients with type 2 diabetes (T2D) with confirmed metformin use and 143 nondiabetic controls, annotating 614 metabolites. Patients were stratified by plasma metformin into sub-therapeutic, therapeutic, and supra-therapeutic groups, and associations were evaluated by multiple linear regression and composite metabolite ranking. Five previously unannotated features were structurally identified as N-lactoyl-amino acids, whose levels correlated strongly with plasma metformin (ρ = 0.42–0.55, P < 0.0001) and increased up to 7.2-fold in the supra-therapeutic group (> 2000 ng/mL). While N-lactoyl-amino acids were consistently detected in the nanomolar range, they still displayed robust and dose-dependent associations with metformin. Broader metabolic changes in T2D included elevated lactate, organic acids, and branched-chain amino acids, together with reduced urea cycle metabolites. Lipidomics showed increases in saturated triacylglycerols and diacylglycerols and decreases in cholesteryl esters, sphingomyelins, and phospholipids. These findings establish N-lactoyl-amino acids as robust, dose-responsive plasma biomarkers of metformin exposure. Despite being up to four orders of magnitude less abundant than their amino acid precursors, they sensitively reflect mitochondrial lactate overflow and pharmacodynamic adaptation, offering objective assessment of adherence.




