From venous blood to VAMS – adaptation of a commercial CE-IVD assay kit for the determination of Tacrolimus, Sirolimus, Everolimus and Cyclosporin A

- Photo: Bioanalytic: From venous blood to VAMS – adaptation of a commercial CE-IVD assay kit for the determination of Tacrolimus, Sirolimus, Everolimus and Cyclosporin A
Background
TDM of immunosuppressants is extremely important topic, especially when it comes to sample collection at home coupled with simple and fast sample preparation and analysis. The aims of the study covered check if optimization of ready-to-use kit for determination of Tacrolimus (TAC), Sirolimus (SIR), Everolimus (EVR) and Cyclosporin A (CSA) in venous blood can be successfully applied for VAMS-collected blood and preliminary venous blood to Mitra® (VB/M) correlation study.
Check the link for pdf of the original poster in the LabRulezLCMS library!
Methods
Sample preparation for venous blood was done according to the procedure provided with the kit (Chromsystems kit: 93000). Sample preparation for Mitra®-collected blood included additional sonication, vortexing and combining of internal standards with extraction buffer. Only reagents supplied by the manufacturer were used. All samples were analyzed on QTRAP 5500+ LC-MS/MS system (SCIEX). Data processing, quantitation and basic statistical analysis was done in SciexOS 3.3 software (SCIEX).
 Bioanalytic: Venous blood (Chromsystems procedure)
Bioanalytic: Venous blood (Chromsystems procedure)
 Bioanalytic: Mitra - optimized procedure based on Chromsystems kit.
Bioanalytic: Mitra - optimized procedure based on Chromsystems kit.
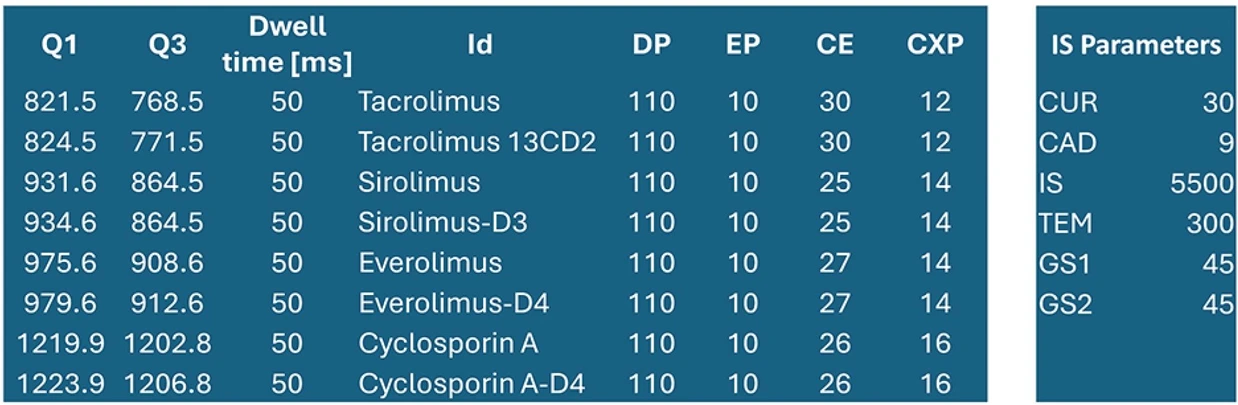 Bioanalytic: Data processing, quantitation and basic statistical analysis was done in SciexOS 3.3 software (SCIEX).
Bioanalytic: Data processing, quantitation and basic statistical analysis was done in SciexOS 3.3 software (SCIEX).Results
The optimized for 10µl and 20µl Mitra® samplers procedure met the validation criteria, including: reproducibility (%CV±15% and accuracy 85-115%), LLOQ (S/N≥10, calibrator 1), linearity (R≥0.995). LOD was not determined, however collected for 10 µl Mitra® sampler data shows that lower that 50 pg/ml concentration for each compound is achieved. The preliminary study of VB/M correlation, included 15 patients per compound where venous blood and 10 µl Mitra® samples were collected at the same time. Obtained VB/M factors were as follows: TAC=0.75, %CV=22.92%, SIR=0.77, %CV=36.10%, CSA=1.15, %CV=19.01%. No patients for EVR were tested during the study.
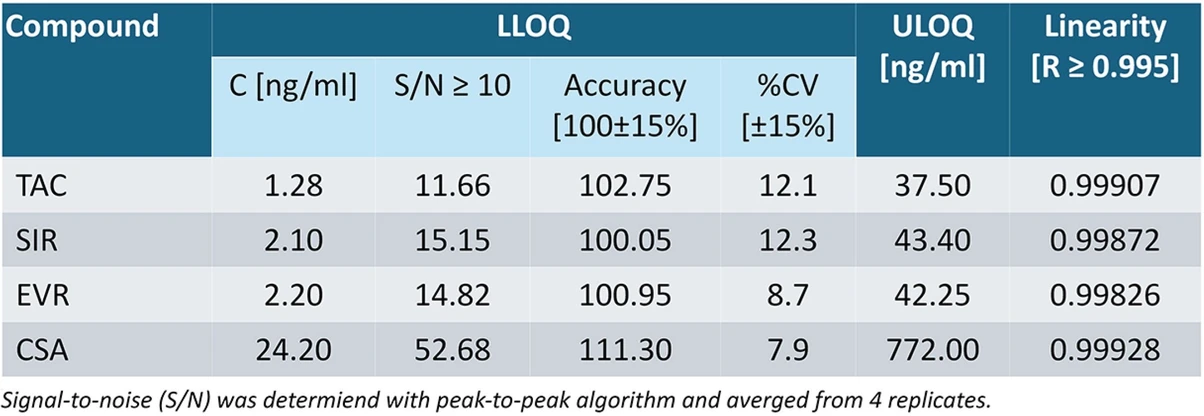 Bioanalytic: Tab. 1. Validation results for collected on 10 microL Mitra sampler lowest analytes concentrations provided with the kit (CAL 1, 4 replicates). LOD was not determined, however assitional analyses showed that lower that 50-100 pg/ml concentration for each compound can be achieved.
Bioanalytic: Tab. 1. Validation results for collected on 10 microL Mitra sampler lowest analytes concentrations provided with the kit (CAL 1, 4 replicates). LOD was not determined, however assitional analyses showed that lower that 50-100 pg/ml concentration for each compound can be achieved.
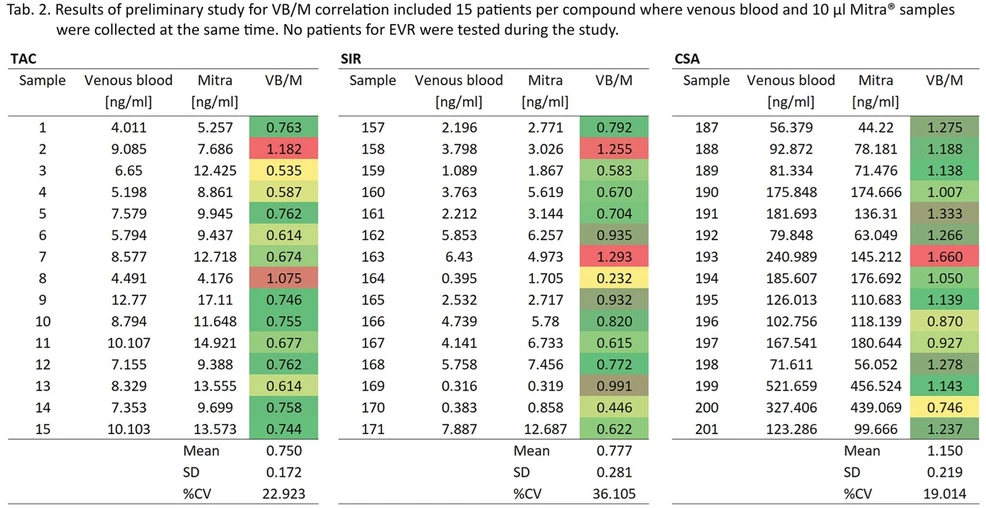 Bioanalytic: Tab. 2. Results of preliminary study for VB/M correlation included 15 paptients par compound where venous blood and 10 microL Mitra samples were collected at the same time. No patients for EVR were tested during this study.
Bioanalytic: Tab. 2. Results of preliminary study for VB/M correlation included 15 paptients par compound where venous blood and 10 microL Mitra samples were collected at the same time. No patients for EVR were tested during this study.
Conclusions
Optimized for VAMS procedure allowed to achieve validation criteria with the use of kit reagents. Additional steps required for Mitra® sample preparation are simple, robust and automation friendly. The VB/M correlations revealed that the capillary blood concentration of TAC and SIR are lower than in venous blood contrary to concentration of CSA. This data may support the fact that TAC and SIR enters erythrocytes in about 90%, while CSA only in 50% and the capillary blood has higher amount of serum fraction. However, due to relatively small number of patients included in the study it needs further investigation on larger group of patients and deeper statistical analysis.
Authors
Rafał Szewczyk1,4; Arkadiusz Kocur2,3, Agnieszka Czajkowska3; Karina Izdebska4; Maciej Stopa1,4. 1LabExperts Sp. z o.o., Gdańsk, Poland; 2Department of Drug Chemistry, Pharmaceutical and Biomedical Analysis, Faculty of Pharmacy, Medical University of Warsaw, Warsaw, Poland; 3Therapeutic Drug Monitoring, Clinical Pharmacokinetics and Toxicology Laboratory Unit, Department of Clinical Biochemistry, The Children’s Memorial Health Institute, Warsaw, Poland; 4Bioanalytic Sp. z o.o., Gdańsk, Poland