Effect of Harvest Time on the Biofunctional Properties of Schizonepeta tenuifolia Briq
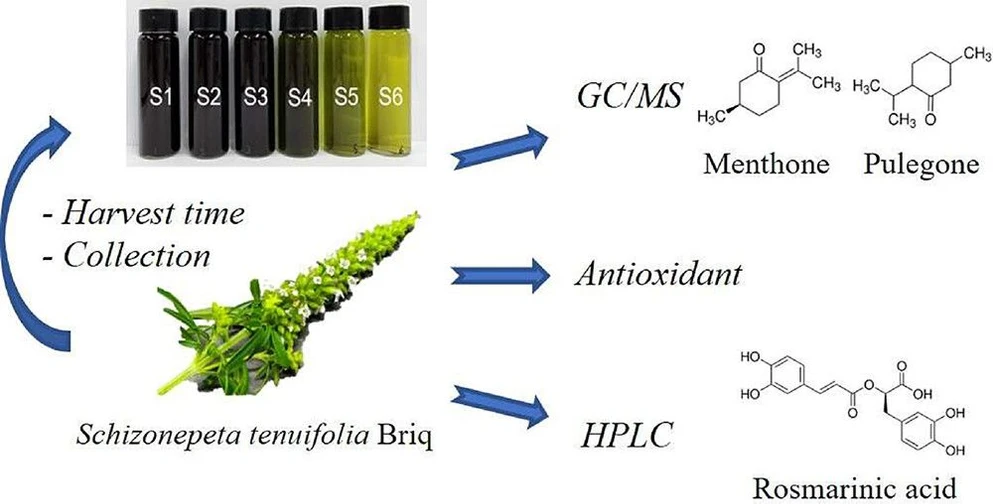
ACS Omega 2025, 10, 49, 60579–60584: Graphical abstract
This study investigates the impact of harvest time on the biofunctional properties of Schizonepeta tenuifolia, a medicinal plant widely used in herbal medicine. Key compounds, including rosmarinic acid, pulegone, and menthone, were quantified using HPLC and GC–MS, while antioxidant activity was assessed using DPPH, ABTS, and FRAP assays.
Results showed that antioxidant capacity and phenolic content varied significantly with growth stage. Rosmarinic acid peaked at stage S5 and strongly correlated with antioxidant activity, while pulegone and menthone reached maximum levels at earlier and later stages, respectively. The optimal harvest periods were identified at stages S4 and S5, maximizing antioxidant potential and overall product quality.
The original article
Effect of Harvest Time on the Biofunctional Properties of Schizonepeta tenuifolia Briq
Yong-Nam Cho, Yun-Sook Kim, Yeong-Bin Kim, Sang-Eun Lee, Min-Ji Gang, Se-Hee Jeon, Jae-Young Heo, and Sung-Tae Lee*
ACS Omega 2025, 10, 49, 60579–60584
https://doi.org/10.1021/acsomega.5c08375
licensed under CC-BY 4.0
Selected sections from the article follow. Formats and hyperlinks were adapted from the original.
S. tenuifolia is a perennial plant from the Lamiaceae family, commonly used for both edible and medicinal purposes across Asia. Reaching a height of 60–150 cm, S. tenuifolia is a perennial plant characterized by a strong, woody stem and multiple branches. Its base is slightly quadrangular, appearing superficially as an obtuse quadrilateral with shallow grooves and a light covering of short hair.
The dried aerial parts of S. tenuifolia are used in herbal medicine to treat various skin diseases. (1,2) They also exhibit anti-inflammatory, antipruritic, insecticidal, and antiviral properties. (3) The main chemical components of S. tenuifolia are volatile oils, such as monoterpenoids (including pulegone, limonene, and menthone) (4) and compounds (Figure 1), such as phenolic compounds and glycosides. Pulegone, which is used in aromatherapy for flavoring and perfume, is a natural monoterpene obtained from the volatile oils of various plants. (4) However, reports suggest that it is a potent hepatotoxin whose metabolism generates p-cresol. (5) The mint-flavored natural monoterpene menthone has been widely used as a flavoring agent or perfume in the cosmetic, drug, and food industries. It is also used as a cooling agent, counterirritant for pain relief, and antidepressant-like substance. (6)
All plants produce a variety of secondary metabolites to adapt to their environment. One of the most important groups of these metabolites are phenolic compounds, which are characterized by one or more aromatic rings (C6) containing one or more hydroxyl groups. (7) They play a role in various functions including plant growth, development, and defense. Phenols act as precursors to other important bioactive compounds commonly used in the pharmaceutical, cosmetic, and food industries. (8)
Current trends in the food industry favor natural biofunctional compounds due to the increased demand and interest in health food. Therefore, S. tenuifolia is an excellent candidate. Traditionally, S. tenuifolia has been used as an ingredient in oriental medicine as well as in tea and soups. However, they contain volatile oils and are greatly affected by climate, cultivation conditions, and harvesting time. The objective of this study was to compare the biofunctional properties of S. tenuifolia, such as antioxidant activities, total phenolic content, and individual phenolic compounds at different harvest times to optimize its production in the food industry.
Materials and Methods
HPLC Analysis of Rosmarinic Acid and Luteolin
HPLC analysis was performed using a system from SHIMADZU Corporation (Japan) with a UV–VIS detector (SPD-20A), a pump (LC-20AD), a column oven (CTO-20A), and LCsolution software. The contents of rosmarinic acid (RA) and luteolin (LU) in the S. tenuifolia extract were quantified based on the method of Wang et al., (12) with slight modifications. The mobile phase consisted of water with 0.1% acetic acid (solvent A) and methanol (solvent B). Solvent B was changed as follows: 0 min, 80% B; 10 min, 40% B; 15 min, 20% B; 25 min, 10% B; and 25–40 min, 80% B. The total running time was 40 min, with a flow rate of 0.5 mL/min. A YMC-Pack Pro C18 RS column (250 × 4.6 mm, 5.0 μm particle size) was used at 30 °C. Before injection, the extracts were filtered using a 0.45-μm syringe filter (Advantec), and the absorbance was measured at 330 nm. RA and LU were identified by their respective peaks and retention times with reference to standard compounds. Calibration curves were prepared using RA (y = 3.970148e-005x + 10.26398, R2 = 0.9992) and LU (y = 3.339664e-005x + 3.114092, R2 = 0.9999) standards, and the phenolic compounds in the extracts were expressed as mg/g DW.
Gas Chromatography–Mass Spectrometry (GC–MS) Analysis
A 1 g sample of S. tenuifolia was placed in a 50 mL conical tube, and 20 mL of chloroform was added. The sample was soaked at room temperature for 2 h. After centrifugation, 1 mL of the extract was filtered through a 0.2-μm syringe filter and used for the analysis. GC–MS analysis was performed using an Agilent 7890A gas chromatograph (Agilent Technologies, USA) with a 5975C mass selective detector and autosampler (split ratio: 50:1), controlled by Agilent Chemstation software. The analysis was conducted on a DB-5MS column (30 × 250 μm × 0.25 μm film) with helium as the carrier gas at 1 mL/min. The temperature program was as follows: hold at 50 °C for 4 min, increase to 220 °C at 4 °C/min with a 2 min hold, and then increase to 280 °C at 20 °C/min with a 15 min hold. The injection temperature was 250 °C, with the transfer line at 280 °C and the ion source at 230 °C. The mass spectrometer was operated in electron impact mode at 70 eV with selected ion monitoring (SIM). Pulegone and menthone were used as standards (y = 1069.8x – 6596.1, R2 = 0.9991) and expressed as mg/kg.
Results and Discussion
GC–MS Analysis
The two compounds identified using the SIM mode are presented in Table 4. In this study, the two major components, pulegone and menthone, were found to constitute 65.6 and 4.35% of the total peak area, respectively. Figure 4 illustrates the bioactive constituents identified in S. tenuifolia. The pulegone content increased from the growing stage (S1) to the spike formation stage (S3) and decreased from the full bloom stage (S4) to the fruiting stage (S6), with the maximum value of 8.55 ± 0.55 mg/g observed at S3. Conversely, the menthone content consistently increased from the growing stage (S1) to the fruiting stage (S6), reaching 1.15 ± 0.43 mg/g at the fruiting stage (S6), with a particularly sharp increase observed from the fruiting formation stage (S5) to the fruiting stage (S6). Overall, the results suggest that the optimal harvest time for S. tenuifolia, based on the content of its major target compound, pulegone, is from the spike formation stage (S2–S3), during which the pulegone content was 1.7–3.0 times higher than at other stages. However, previous studies indicated that the optimal harvesting time for S. tenuifolia was at the spike formation stage. Although the content of pulegone showed a slight increasing trend during harvesting times, the menthone content tended to decrease over the same period. (4) Notably, the quality of S. tenuifolia may have been influenced by variations in the cultivars and growing conditions. Several studies have shown that different cultivars and growing conditions significantly affect crop performance. (20,21)
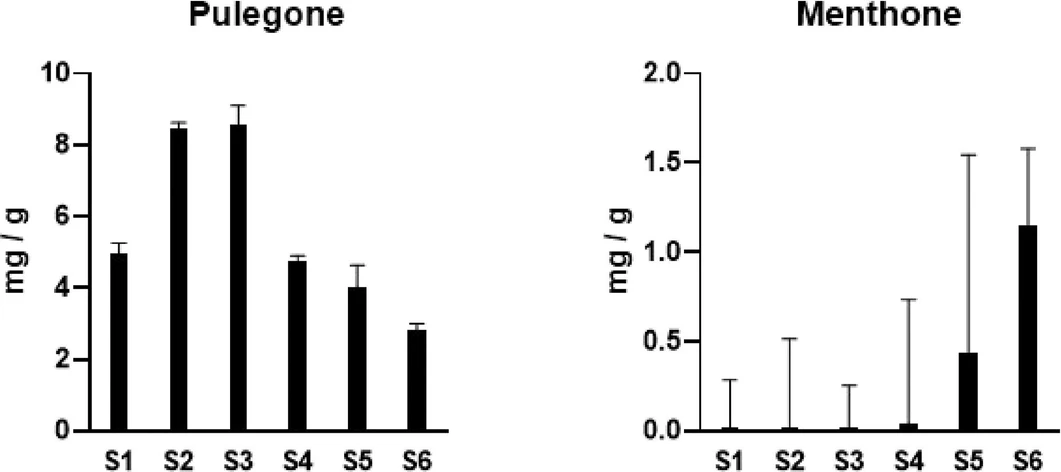 ACS Omega 2025, 10, 49, 60579–60584: Figure 4. GC–MS analysis of pulegone and menthone contents in S. tenuifolia extracts.
ACS Omega 2025, 10, 49, 60579–60584: Figure 4. GC–MS analysis of pulegone and menthone contents in S. tenuifolia extracts.
Conclusions
This study demonstrates that the functional properties of S. tenuifolia, such as antioxidant activity and phenolic content, are significantly influenced by the harvesting time. Previous studies on S. tenuifolia or related Lamiaceae species have mainly focused on evaluating the antioxidant activity of crude extracts or quantifying a limited number of compounds at a single harvest stage. In contrast, our work systematically compared multiple functional parameters across five distinct growth stages, integrating changes in phenolic compounds (especially, rosmarinic acid), volatile monoterpenes (pulegone and menthone), and antioxidant activities. The best harvesting time for ABTS, FRAP, and TPC was S4, compared to other stages, and for DPPH it was S6. The contents of RA and LU in S. tenuifolia extracts showed the highest content of RA at S5. RA was strongly correlated with DPPH radical scavenging (R2 = 0.877–0.904), ABTS radical scavenging (R2 = 0.890–0.907), and FRAP (R2 = 0.964–0.980). The optimal harvesting times for pulegone and menthone are likely during S3 to S4, with menthone undergoing an approximately 15-fold increase from S3 to S4. Overall, the results suggest that the optimal harvesting time for maximizing these functional properties, while also maintaining high levels of pulegone and menthone, is during S4 and S5, with a marked increase in the antioxidant activity and phenolic content. By identifying the best harvesting window, producers can enhance the quality and health benefits of S. tenuifolia-based products, improving their effectiveness in foods, herbal remedies, and dietary supplements.