Oligos Made Easy - Part 4: HILIC

KNAUER: Oligos Made Easy - Part 4: HILIC
Why Consider HILIC in Modern Oligonucleotide Analysis?
Oligonucleotides play a central role in gene therapies, molecular diagnostics, and cutting-edge biomedical research. Before these molecules reach application, however, they must be carefully characterized to confirm purity, identity, and performance.
Analyzing therapeutic oligonucleotides is far from straightforward. Their polarity, structural diversity, and modifications demand highly selective and MS-compatible separation methods. Although ion-pair reversed-phase LC (IP-RP-LC) remains the established standard for LC-MS workflows, hydrophilic interaction liquid chromatography (HILIC) is rapidly emerging as a powerful complementary technique.
When IP-RP approaches reach practical or performance limits, HILIC offers an attractive alternative, delivering strong retention and efficient separation of highly polar oligonucleotides — without relying on ion-pairing reagents.
How Does HILIC Separate Oligonucleotides?
HILIC essentially reverses conventional reversed-phase logic. It combines:
- A polar stationary phase
- An organic-rich mobile phase
In this configuration, the stationary phase retains a thin, water-enriched layer on its surface. Polar analytes — such as oligonucleotides — partition into this aqueous layer while the acetonitrile-rich mobile phase passes through.
This mechanism allows:
- Retention of short or highly polar oligonucleotides
- Separation of modified sequences
- LC-MS compatibility without ion-pairing reagents
The key to performance lies in carefully tuning the parameters that govern HILIC selectivity and retention.
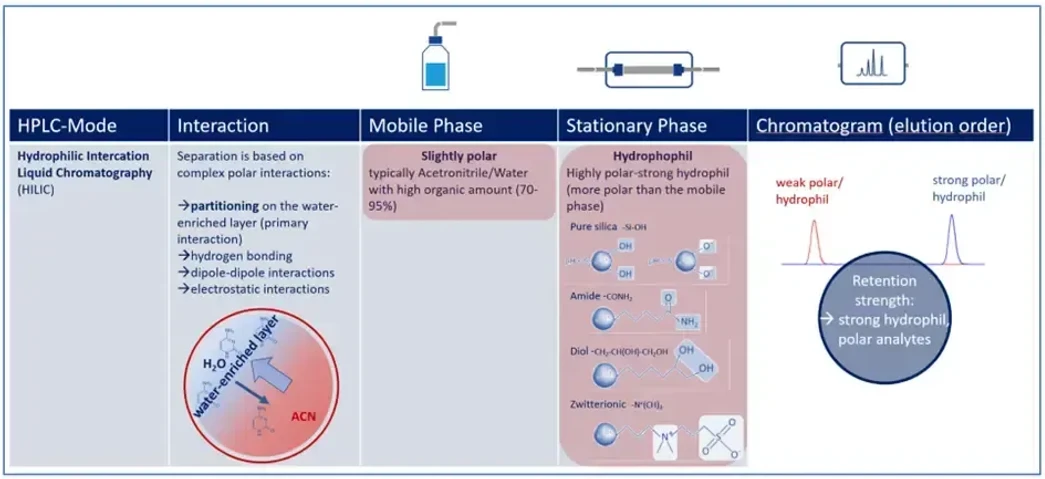 KNAUER: Figure 1 - Key Parameter of HILIC Mode.
KNAUER: Figure 1 - Key Parameter of HILIC Mode.
What Controls Retention in HILIC?
Several interaction mechanisms contribute to analyte behavior in HILIC:
Hydrophilic Partitioning – The Primary Mechanism
Polar molecules distribute into the thin water layer formed on the stationary phase surface. The greater the polarity of the analyte, the stronger its affinity for this layer, leading to increased retention.
Hydrogen Bonding – Fine-Tuning Effects
Functional groups such as –OH, –NH₂, and –COOH can form hydrogen bonds with the stationary phase, subtly influencing retention time and selectivity.
Electrostatic Contributions – The Role of Charge
Some HILIC materials (e.g., bare silica) exhibit surface charges. Depending on mobile phase pH and buffer composition, electrostatic interactions can either enhance or suppress analyte retention.
Dipole and Ion–Dipole Interactions – Added Selectivity
Highly polar or zwitterionic compounds — including phosphorylated species and sugars — may engage in dipole–dipole or ion–dipole interactions, further refining separation behavior.
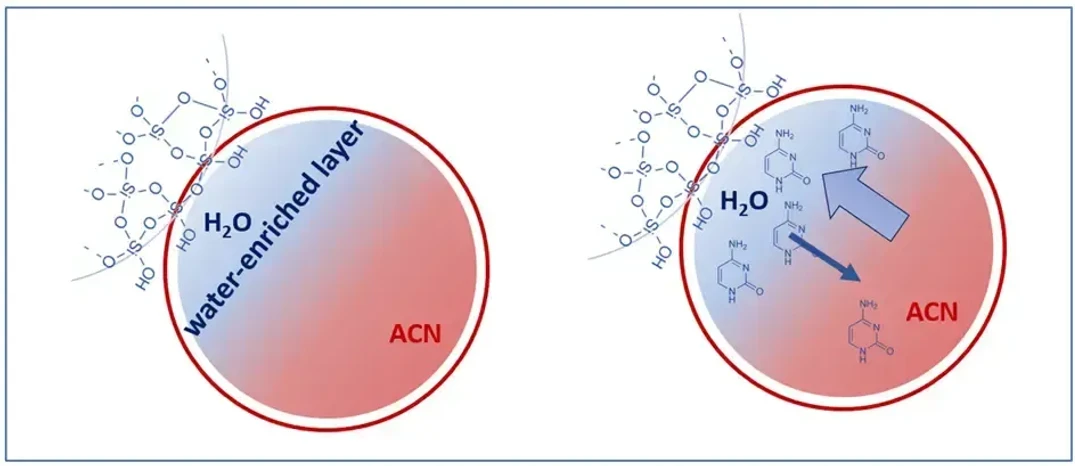 KNAUER: Figure 2 - Hydrophilic Partitioning on Water-enriched Layer.
KNAUER: Figure 2 - Hydrophilic Partitioning on Water-enriched Layer.
How to Develop a HILIC Method for Oligonucleotides
Like any chromatographic method, successful HILIC development follows a structured approach:
Step 1: Define the Analytes
Understand oligonucleotide length, polarity, charge, and modifications.
Step 2: Select a Suitable Column
Choose a polar stationary phase (e.g., amide or zwitterionic) appropriate for the oligonucleotide’s chemical properties.
Step 3: Optimize the Mobile Phase
- Start with 70–95% acetonitrile
- Use a volatile buffer (e.g., ammonium formate or ammonium acetate, typically 5–20 mM)
- Adjust pH (commonly between 6–8, with testing up to pH 9 if column stability allows)
Small pH shifts can significantly alter selectivity and MS response.
Step 4: Design the Gradient
Employ a gradient that increases water content to modulate retention and separate oligonucleotides by size or chemical modification.
Step 5: Match Injection Conditions
Samples should be injected in a solvent similar to the starting mobile phase (high organic content) to prevent peak distortion.
Step 6: Evaluate Performance
Assess:
- Resolution
- Retention time stability
- Peak shape
- Reproducibility
Balancing separation performance with MS ionization efficiency is essential.
 KNAUER: Table 1 - HILIC Method Step Procedure for Oligos.
KNAUER: Table 1 - HILIC Method Step Procedure for Oligos.
Practical Tips for High-Performance HILIC
Maintain a Stable Water Layer
A minimum of ~5% water is necessary at the start of the run. Too little water can eliminate retention entirely. Adjust water content carefully to fine-tune retention.
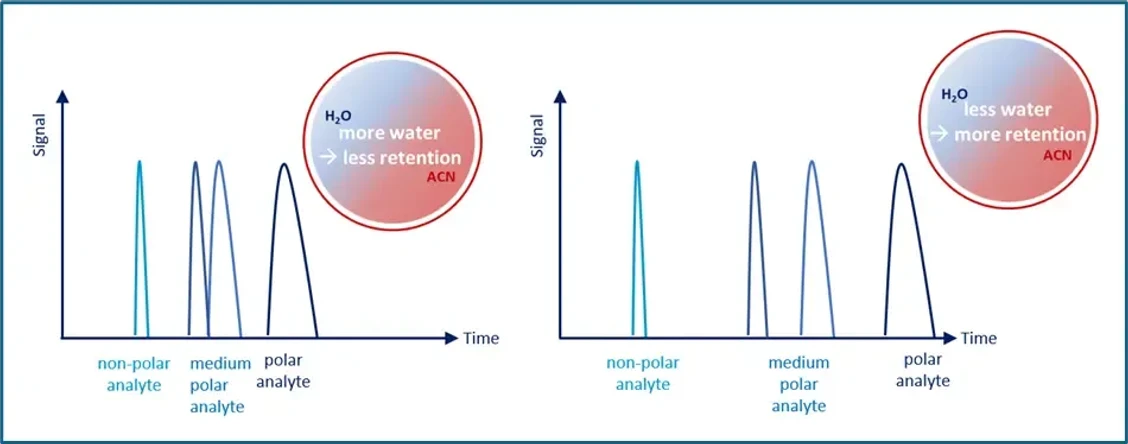 KNAUER: Figure 3 - Water Adjustment Principle for HILIC.
KNAUER: Figure 3 - Water Adjustment Principle for HILIC.
Use High Organic Content
HILIC typically operates best with 70–95% acetonitrile. Polar analytes are retained more effectively under these conditions.
Include a Volatile Buffer
Use low concentrations (5–20 mM) of ammonium formate or acetate to:
- Stabilize pH
- Reduce peak tailing
- Maintain MS compatibility
Control Injection Solvent
Avoid high-water sample solvents. They disrupt the partitioning mechanism and distort peaks.
Allow Proper Column Equilibration
HILIC columns require longer equilibration times than reversed-phase columns — typically 10–20 column volumes after major gradient or solvent changes.
Consider Temperature
Operating at 30–50 °C can:
- Improve peak shape
- Reduce mobile phase viscosity
- Stabilize retention behavior
From Alternative to Essential? Final Perspective
HILIC-MS is steadily advancing as a strong, ion-pair-free option for oligonucleotide analysis. While IP-RP-LC still dominates in sensitivity for many applications, HILIC is closing the gap and offering valuable flexibility — particularly for highly polar or challenging sequences.
Although HILIC follows different chromatographic principles than reversed-phase separations, once optimized, it can deliver robust and highly selective results.
Rather than being a niche method, HILIC is becoming a strategic tool in modern oligonucleotide workflows — especially when MS compatibility and method versatility are priorities.
If you are developing or refining an oligonucleotide LC-MS method, HILIC may be worth integrating into your analytical toolbox.
- For additional information or discussion, feel free to contact: [email protected]
- For direct scientific inquiries, reach out to the author at: [email protected]
Stay tuned for more insights in the “Oligos Made Easy” series.




