Oligos Made Easy - Part 5: SEC
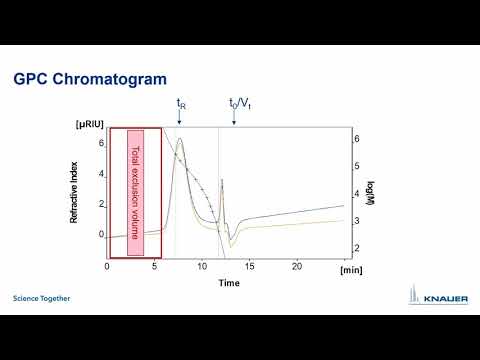
- Photo: KNAUER: Oligos Made Easy - Part 5: SEC
- Video: KnauerHPLC: GPC and SEC theory, principles and parameters
Size Matters: A Practical Guide to Size Exclusion Chromatography for Oligonucleotides
Why Consider SEC in Modern Oligonucleotide Workflows?
Oligonucleotides are now central to advanced therapeutics, molecular diagnostics, and synthetic biology. As their applications expand, so does the need for highly purified and well-characterized products.
Size Exclusion Chromatography (SEC) offers a valuable solution in this evolving landscape. Unlike separation techniques based on charge or hydrophobicity, SEC separates molecules strictly according to their size. This means oligos remain structurally intact during purification while unwanted components — such as salts, truncated strands, aggregates, and small molecular impurities — are removed.
SEC can be applied to smaller oligos, but it is particularly powerful for desalting and buffer exchange. For high-resolution separations and detailed characterization, techniques such as AEX or HILIC are often preferred. Understanding the strengths of SEC allows you to use it effectively within your purification strategy.
How SEC Purifies Oligonucleotides
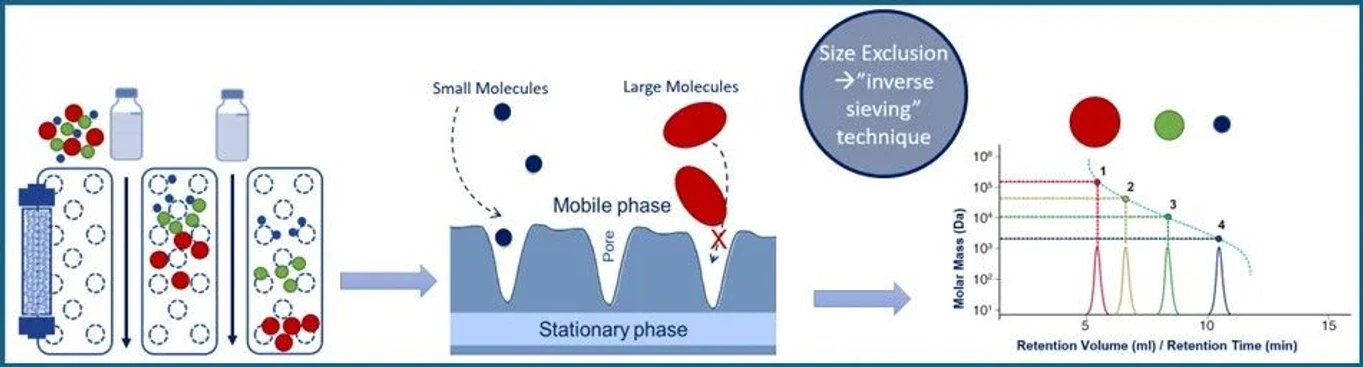
Imagine a chromatographic column filled with microscopic porous beads. These beads act like selective barriers: molecules are separated based on whether they can enter the pores or not.
When a mixture containing oligonucleotides and impurities passes through:
- Large oligos are excluded from the pores and move quickly around the beads.
- Smaller molecules enter the pores and travel a longer path.
- Medium-sized species fall somewhere in between.
As a result, the target oligonucleotide elutes first, separated from salts, degraded fragments, and other small contaminants.
Importantly, SEC operates under native conditions. There are no strong interactions, no harsh gradients, and no disruption of secondary structures. Separation is governed purely by molecular size.
 KNAUER: Figure 1 - SEC Principle.
KNAUER: Figure 1 - SEC Principle.
The Core Principle: Size Dictates Elution
In SEC, molecular dimensions determine migration speed:
- Large oligos cannot penetrate the pores and therefore elute first.
- Small impurities such as salts and solvent residues enter the pores and elute last.
- Intermediate species, including truncated oligos, elute between the two extremes.
This mechanism makes SEC ideal for allowing your target oligo to “take the fast lane” while smaller contaminants are retained longer within the porous matrix.
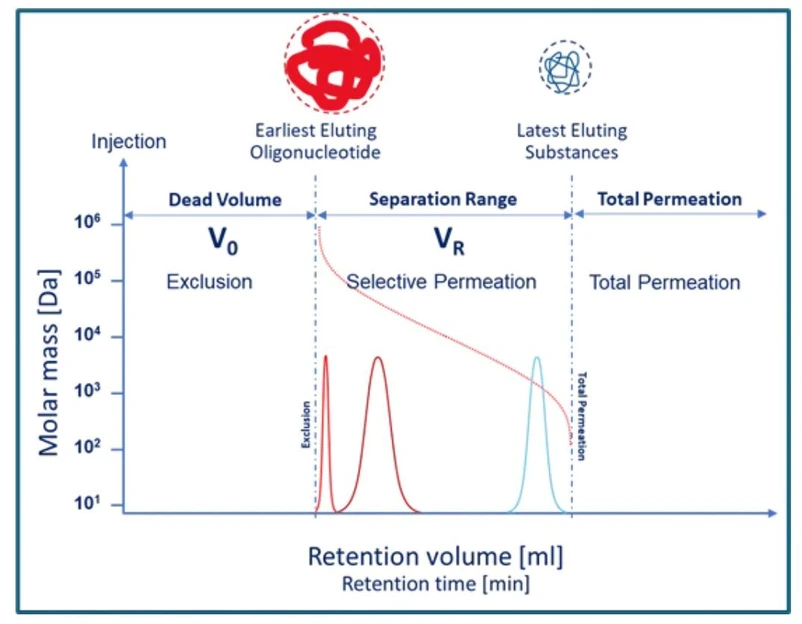 KANUER: Figure 2 - SEC Elution
KANUER: Figure 2 - SEC Elution
Step-by-Step Guide to Oligo Purification Using SEC
1. Selecting the Appropriate Column
Typical oligonucleotides range from 10–50 nucleotides (~3–15 kDa).
Recommended pore sizes:
- 100–150 Å for short oligos (10–25 nt)
- 200–300 Å for longer strands (25–50 nt) or aggregates
Columns packed with high-quality SEC media such as AppliChrom® or equivalent materials are well suited for these applications.
2. Preparing the Sample
- Allow complete dissolution (overnight if necessary).
- Filter samples to prevent column blockage.
- Maintain moderate concentrations (e.g., 0.1–1 mg/mL) for optimal separation.
3. Choosing the Mobile Phase
SEC for oligonucleotides typically employs simple aqueous buffers:
- 50 mM phosphate or Tris
- 100–150 mM NaCl (to reduce non-specific interactions)
Avoid detergents and organic modifiers that may compromise column stability.
After buffer use, flush with water (optionally containing sodium azide) to maintain column integrity.
4. Optimizing Flow Rate
- Analytical columns: 0.2–0.5 mL/min improves resolution.
- Preparative runs: higher flow rates can be used cautiously, balancing speed and separation quality.
Always monitor system backpressure.
5. Sample Loading
Avoid overloading. Excess injection volumes compromise resolution.
Confirm the resin’s loading capacity before scaling up.
6. Fraction Collection
Monitor UV absorbance at 260 nm.
Collect early-eluting peaks corresponding to the target oligo.
Later fractions generally contain salts and smaller impurities.
7. Purity Verification
Confirm purity using complementary analytical methods such as:
- AEX
- IP-RP
- HILIC HPLC
Critical Column Parameters That Influence Performance
Pore Size
Pore dimensions determine the effective separation range. Proper pore selection ensures clean size-based discrimination between oligos and contaminants.
Inert Surface Chemistry
The ideal SEC matrix should be non-interactive:
- No ionic interactions
- No hydrophobic adsorption
- No secondary retention effects
Separation should rely solely on molecular size, not chemical affinity.
Mechanical and Chemical Stability
Reliable SEC gels maintain:
- Structural integrity under pressure
- Stability in various aqueous buffers
- Performance at elevated temperatures
Abrupt changes in flow rate, temperature, or solvent composition should be avoided.
Hydrophilicity
SEC media are typically hydrophilic and compatible with aqueous systems. Common materials include:
- Crosslinked dextran
- Agarose
- Polyacrylamide
- Hydroxylated methacrylate
These materials are well suited for oligonucleotide purification.
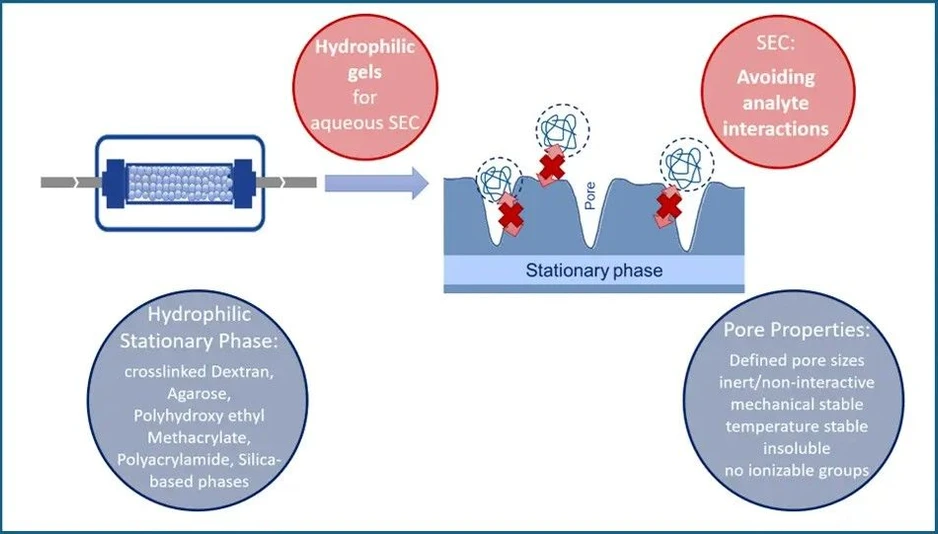 KNAUER: Figure 3 - SEC Column Gel Parameter Characterization
KNAUER: Figure 3 - SEC Column Gel Parameter Characterization
Practical Optimization Tips
- Desalting pre-treatment: Avoid redundant desalting if the sample is salt-rich.
- Temperature control: Maintain consistent room temperature or use a column thermostat.
- pH range: Keep between 6.5–8.0 to protect both oligos and gel stability.
- Buffer exchange: Use your final formulation buffer as the mobile phase when possible.
- Injection volume (analytical): <2–5% of column volume.
- Injection volume (preparative): 5–10%, depending on resin capacity.
- Pressure management: Avoid sudden changes to prevent bed collapse.
- Fraction collection: Base cuts on peak shape and symmetry, not retention time alone.
- Column care: Use filtered samples, flush properly, store in preservative (e.g., 20–30% ethanol), and monitor performance routinely.
Proper maintenance ensures reproducible, long-term performance.
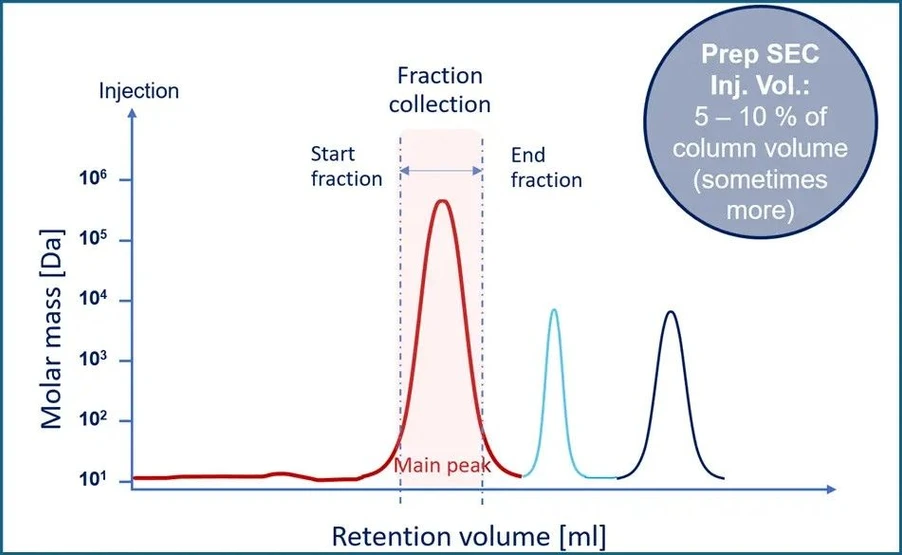 KNAUER: Figure 4 - Preparative SEC with Fraction Collection
KNAUER: Figure 4 - Preparative SEC with Fraction Collection
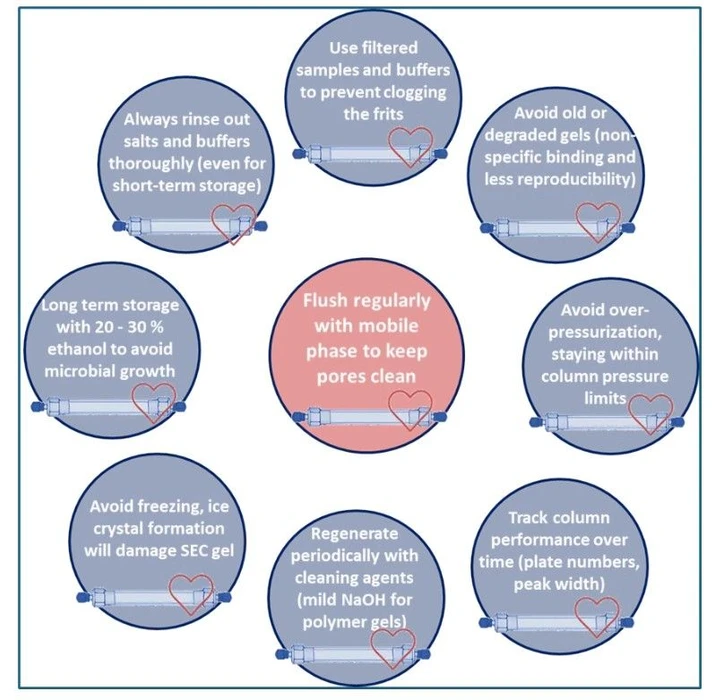 KANUER: Figure 5 - SEC Column Maintenance
KANUER: Figure 5 - SEC Column Maintenance
Strengths and Limitations in Practice
SEC is widely appreciated for its simplicity and gentle operation. It does not require gradients or complex additives and maintains oligonucleotide integrity.
However, it is not universally superior.
Where SEC Excels:
- Desalting
- Buffer exchange
- Removal of small molecules
- Rapid quality control checks
- Aggregate analysis
Where Other Methods May Be Better:
- High-resolution impurity profiling
- Detailed structural characterization
- Separation of closely related sequence variants
Techniques such as AEX, IP-RP, or HILIC frequently provide higher resolution for demanding analytical tasks.
Final Takeaway
Size Exclusion Chromatography is a dependable and versatile tool in oligonucleotide purification. While it may not replace higher-resolution chromatographic methods, it remains an essential component of efficient purification workflows.
The key is not choosing one method exclusively — but knowing when SEC is the right solution and when to complement it with alternative techniques for optimal results.
If you're planning to set up or optimize your oligonucleotide workflow, feel free to contact us at [email protected]. Stay tuned for more exciting insights into the Oligonucleotide world in our “Oligos Made Easy” series.




