Oligos Made Easy - Part 6: Column Navigator

KNAUER: Oligos Made Easy - Part 6: Column Navigator
How to Choose the Right Column for Oligonucleotide Analysis
Finding the Best Column for Your Oligonucleotides
Selecting an appropriate chromatographic column for oligonucleotide analysis can be challenging. These molecules are long, highly charged, and often require specialized separation strategies. With multiple chromatographic techniques available, choosing the most suitable approach for your application is not always straightforward.
However, understanding the characteristics of oligonucleotides and the analytical requirements of your workflow can significantly simplify the process.
Why Are Oligonucleotides Difficult to Separate?
Over the past several years, oligonucleotides have become increasingly important in molecular biology, biotechnology, and therapeutic research. Their growing role in regulated environments, including pharmaceutical development, means that precise and reliable analytical methods are essential.
From a chromatographic perspective, oligonucleotides present several challenges.
These molecules are typically highly polar and strongly charged, consisting of phosphodiester or phosphorothioate backbones. During synthesis, numerous closely related impurities can be generated. These include fragments that differ slightly in chain length (shortmers or longmers), as well as molecules with minor structural variations such as single-base substitutions or functional group modifications.
In addition, oligonucleotide samples frequently contain residual reagents, salts, and by-products from synthesis. These components can overload chromatographic columns and negatively affect separation efficiency.
For analytical laboratories, this means that a suitable column must deliver:
- High resolution between closely related structures
- Symmetrical and well-defined peaks
- Fast analysis times
- Compatibility with mass spectrometry (MS) detection
At the same time, the method must provide reliable reproducibility and minimize sample loss during elution or desalting.
Understanding these analytical requirements is the first step toward selecting the most appropriate chromatographic solution.
Which Chromatographic Approach Should You Choose?
Separating oligonucleotides is inherently complex because many compounds differ only slightly in structure, sometimes by a single nucleotide or modification.
Different chromatographic techniques are therefore used depending on the analytical objective.
For purification of synthesized strands, preparative liquid chromatography or size exclusion chromatography (SEC) is typically employed.
For analytical characterization, UHPLC coupled with mass spectrometry is widely used because it enables both qualitative and quantitative analysis of products and impurities.
Before purification or analysis, samples may also require desalting, which can be achieved efficiently using dedicated SEC desalting cartridges to remove interfering salts.
Even when the general purpose—purification or analysis—is known, it is still necessary to decide how detailed the separation needs to be and whether robustness or analytical depth should take priority.
“Clearly defining your chromatographic goals is the first step toward selecting the right separation method.”
Dr. Ulrike Krop, Head of Applications & Academy, KNAUER
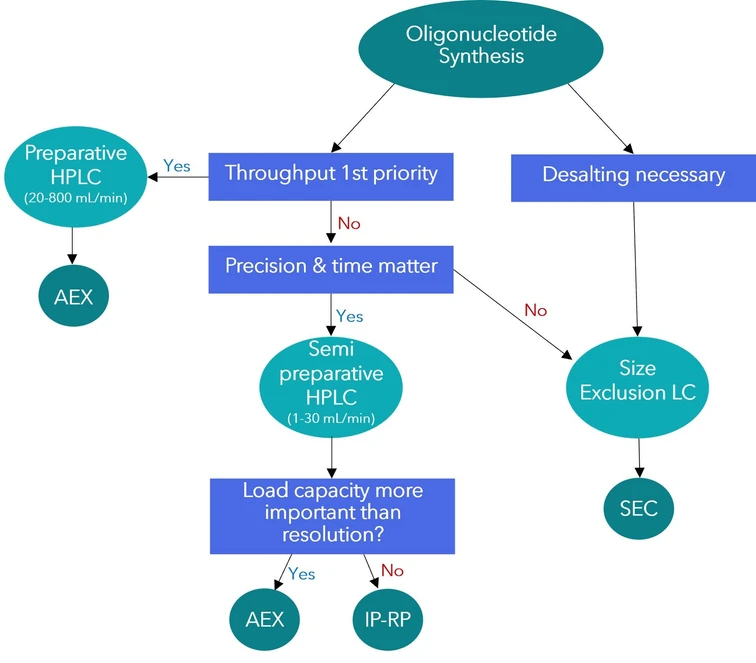 KNAUER: Figure 1: Decision tree - Which separation technology for your purification task?
KNAUER: Figure 1: Decision tree - Which separation technology for your purification task?
Choosing a Method for Oligonucleotide Purification
When the goal is purification, the amount of material to be processed plays a key role.
If only small quantities of oligonucleotides need to be purified, columns with smaller internal diameters and lower flow rates are typically sufficient.
For applications where high resolution is required, ion-pair reversed-phase chromatography (IP-RP) can be an effective option.
Alternatively, laboratories that prioritize environmentally friendly eluents, cost efficiency, and high loading capacity may prefer anion exchange chromatography (AEX) for preparative separations.
If the objective is mainly to separate smaller fragments or remove residual synthesis reagents, size exclusion chromatography (SEC) provides a simple and environmentally friendly solution.
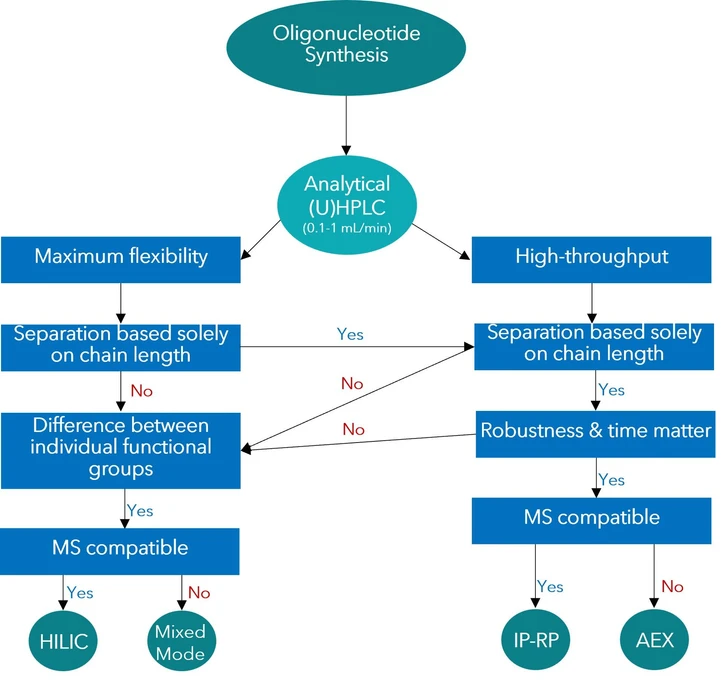 KNAUER: Figure 2: Decision tree – What are your priorities for your analysis?
KNAUER: Figure 2: Decision tree – What are your priorities for your analysis?
Selecting an Analytical Strategy
In analytical workflows, the decision often comes down to a balance between maximum resolution and operational robustness.
High-throughput laboratories that routinely perform similar analyses and require stable long-term operation typically choose between IP-RP and AEX methods.
Among these approaches, ion-pair reversed-phase chromatography has become widely established due to its excellent efficiency and resolution. In addition, IP-RP methods generally offer good compatibility with mass spectrometry, making them suitable for detailed impurity profiling.
Columns specifically designed for oligonucleotide separations, such as Sepapure oliGO columns, are optimized for this application and provide reliable performance across a wide range of workflows.
Methods for Advanced Research Applications
In research and development settings, analytical flexibility and sensitivity may be more important than routine efficiency.
For example, if the goal is to identify minor differences in side-chain modifications between two otherwise identical oligonucleotides, techniques such as HILIC (hydrophilic interaction chromatography) or mixed-mode chromatography can be particularly useful.
These methods provide additional separation mechanisms and allow extensive method optimization. Depending on the chosen eluents, they can also remain compatible with mass spectrometric detection.
 KNAUER: Benefits and Limitations at a Glance
KNAUER: Benefits and Limitations at a Glance
Do You Need Bioinert Hardware?
Bioinert chromatographic hardware is commonly associated with protein purification, where metal surfaces can interact with amino acid residues such as histidine or cysteine and alter protein structure.
To prevent these interactions, chromatographic systems may use passivated surfaces, titanium components, or glass-lined flow paths. However, these solutions can significantly increase costs.
For oligonucleotide separations, the benefits of bioinert hardware are less clear.
“I have performed many analyses with different oligonucleotides and have not observed significant differences between bioinert and stainless steel hardware. Only in extremely fast UHPLC runs—around three minutes—might this become relevant.”
Marvin Schaek, Product Manager Oligosynthesizer, KNAUER & XNAPharma GmbH
Since oligonucleotide structure is generally less sensitive to conformational changes than proteins, stainless steel hardware often performs adequately.
However, in cases where phosphorothioate backbones are present, interactions with metal surfaces may occur. Some studies suggest that column passivation can improve peak shape in very fast UHPLC separations, although the impact in typical 10-minute methods appears minimal.
Final Thoughts
Anyone working with oligonucleotides will inevitably encounter the need for purification and high-quality chromatographic analysis.
Fortunately, a variety of chromatographic techniques are available, allowing scientists to tailor their workflows to specific analytical goals. By carefully evaluating sample characteristics, desired resolution, and operational priorities, laboratories can select the most appropriate column and separation method.
For IP-RP workflows ranging from UHPLC to semi-preparative scale, columns such as Sepapure oliGO provide a reliable and cost-effective option specifically designed for oligonucleotide applications.
If you are planning to develop or improve your oligonucleotide workflow, feel free to contact the KNAUER team for further guidance.




