Characterization of Asian lacquers by atmospheric solids analysis probe high resolution tandem mass spectrometry coupled with cyclic ion mobility separation
Journal of Cultural Heritage, 77, 2026, 187-196: Fig. 1. Selected objects from each geographical area (details of the objects are given in Table 1 and Supplementary Material A) and Fig. 3. Fragmentation spectra of heptadecylcatechols possessing 0-3 double bonds, (MS/MS; m/z of parent ions are: a) 342.2559, b) 344.2715, c) 346.2872, d) 348.3028).
This study introduces atmospheric solids analysis probe high-resolution tandem mass spectrometry with cyclic ion mobility as a novel approach for characterizing Asian lacquers. Applied to 27 historical samples from Japan, China, Vietnam, and Myanmar, the method enabled classification by geographic origin based on phenol and benzenediol derivatives.
In addition to lacquer identification, the technique simultaneously detected pigments such as arsenic sulfides and associated resins, providing comprehensive chemical insight. This approach expands analytical capabilities for cultural heritage studies, supporting restoration, conservation, and art historical research.
The original article
Characterization of Asian lacquers by atmospheric solids analysis probe high resolution tandem mass spectrometry coupled with cyclic ion mobility separation
Vojtěch Zemek, Radek Ryšánek, Petra Krejčí, Lukáš Kučera, Jana Nádvorníková, Adéla Tůmová, Helena Heroldová, Adriana Stříbrná, Petr Bednář
Journal of Cultural Heritage, 77, 2026, 187-196
https://doi.org/10.1016/j.culher.2025.11.012
licensed under CC-BY 4.0
Selected sections from the article follow. Formats and hyperlinks were adapted from the original.
In recent years, an expansion of ambient pressure ionization mass spectrometry in various areas is observed [24,25]. Atmospheric solids analysis probe mass spectrometry (ASAP-MS) is a powerful technique for direct analysis of even very complex samples, such as polymers [26], foods [27], or petrochemical sources and products [24]. ASAP ionization technique is a modification of atmospheric pressure chemical modification (APCI). In conventional arrangement, the sample is attached to a sampling probe tip (a standard melting point capillary is inserted into powder or liquid sample that sticks to its surface) and the probe is consequently placed into the ion source (commonly adapted ESI or APCI source). The analytes are released into the gas phase using hot gas (nitrogen) and ionized in the corona discharge. The formed ions (quasimolecular ions, radical cations or radical anions) are then transferred to the evacuated part of mass spectrometer [24,25,28]. Note that especially at higher vaporization temperatures a portion of the analytes is pyrolyzed and a mixture of parent ions and pyrolysis product ions is commonly observed in the ASAP-MS spectra [29]. The main feature of this direct MS mode is a fast detection of the formed products (including those with a higher mass and perhaps a shorter lifetime). In contrast, due to chromatographic separation in py-GC/MS, only relatively stable and volatile products are detected (the chromatographic step provides an additional selectivity here). In addition, electron impact ionization produces significantly different spectra profiles compared to the atmospheric pressure chemical ionization ongoing in the ASAP source. Both techniques are to a certain extent complementary [24,25,30].
There is a direct possibility of coupling mass spectrometry with ion mobility spectrometry (IMS) to significantly improve the selectivity of analysis (and, to certain extent, to compensate the advantage of the chromatographic step in py-GC/MS while maintaining the direct MS analysis speed). During an ion mobility spectrometry experiment, ions of analytes are flying through the ion mobility cell (e.g., a drift tube or a closed-loop path within a traveling wave ion guide in case of the cyclic ion mobility cell) filled with buffer gas (e.g. helium). In this process, ions collide with the buffer gas. These collisions slow down the ions differently based on their size, shape and charge. Ion mobility spectrometry therefore allows separation of the present analytes in the gas phase according to their collision cross sections (CCS). It also allows separation of isomeric compounds and filtration of the matrix signals, resulting in a higher quality of the MS and MS/MS spectra [[31], [32], [33]].
ASAP-MS has a high potential for the study of protective and decorative layers on the surfaces of historical objects. To the best of our knowledge, however, ASAP-MS and ASAP-IMS-MS have never been used for characterization of lacquers. This paper deals with the development of ASAP-MS method for characterization of Asian lacquers and its application for classification of oriental historical objects.
3. Material and methods
3.3. Instrumentation and experimental setup
Analyses were performed using Waters Cyclic IMS high resolution tandem mass spectrometer with an integrated cyclic ion mobility cell and atmospheric solids analysis probe (ASAP) used as ion source (Waters, Milford, MA, USA). Samples (∼ 0.1–0.5 mg) were inserted into a melting point capillary (G119/32, manufacturer SAMCO, United Kingdom) through an in-lab radially drilled hole (hole diameter: approx. 1 mm; distance from the bottom end approximately 5 mm). Solution of 2′,4′,6′-trihydroxyacetophenone monohydrate (THAP) in acetone was used as a standard for lock mass correction (c = 1 mg.ml-1). The m/z values were calculated as the means from three repeated measurements of appropriate samples. Approx. 1 μl of standard was deposited onto the capillary using a small pipette tip. Following experimental parameters were used: desolvation temperature of 600 °C, source temperature of 100 °C, corona “current” mode (corona current 2.0 μA), cone voltage of 30 V, source offset of 10 V, cone gas flow of 30 l/min, desolvation gas flow of 500 l/min, nebulizer gas pressure of 6 bar and reference capillary voltage of 2.5 kV. As for the ion mobility separation setup, a cyclic ion mobility cell was used in the following sequence: 1. Inject (10 ms), 2. Separate (37 ms), 3. Inject to Pre-Store (0.50 ms), 4. Eject (0.50 ms), 5. Reinject from Pre-Store (0.63 ms), 6. Eject and Acquire (13.20 ms). TW static height was 11 V, TW ramp start height was 15 V and TW ramp end height was 35 V. Acquisition time was 3 min for the MS scan and 1 min for the MS/MS scan used for identification of analytes. In the MS/MS mode, collision energy 30 V was used for pentadecyl and heptadecylcatechols, 35 V for phenols substituted with (CH2)10C6H5 and 40 V for catechols substituted with (CH2)12C6H5 and phenols substituted with CH2CO(CH2)10C6H5, respectively. Scan time 1 s was used for both scans. At the end of each working day, sample V was measured for confirmation that the instrument measured consistently. Values of m/z were calculated by MassLynx software (version 4.2, SCN 1016) after lock mass correction using the signal of protonated molecule of THAP. Theoretical mass of particular analyte was used for lock mass correction in the MS/MS spectra. Centroid top at 80 % and areas of peaks were used for accurate mass and intensity of signals calculation. The maximal accepted difference from theoretical mass (dtm) of signals was 10 mDa. Peak areas were used to compare the abundance of identified analytes among particular samples. Mobilograms from cyclic ion mobility experiments were produced by DriftScope v2.9 software. In mobilograms, spectra were generated by integration at Full Width at Half Maximum (FWHM) of peaks at drift time 1.72 ms.
4. Results and discussion
4.1. Utilization of ASAP-MS
In the ASAP-MS spectra of the studied lacquers, signals of a several compounds were observed (Fig. 2). Signals of radical cations as well as protonated molecules of identified analytes were easily visible. Usually a slightly higher abundance of cation radicals compared to protonated molecules was observed (spectra of all samples are listed in Supplementary material B). The processes observed in the ASAP-MS spectra involve the release of alkyl phenol/alkyl catechol monomer(s) and cleavages of alkyl chain(s) after the carbon linked to the phenol/catechol ring (i.e. cleavage after the first, second and third alkyl carbons) and elimination of water either from polymer or released monomers (the pyrolytic/collision-induced fragmentation processes are discussed below in detail). The analysis of provided samples revealed the most important markers of specific types of lacquers.
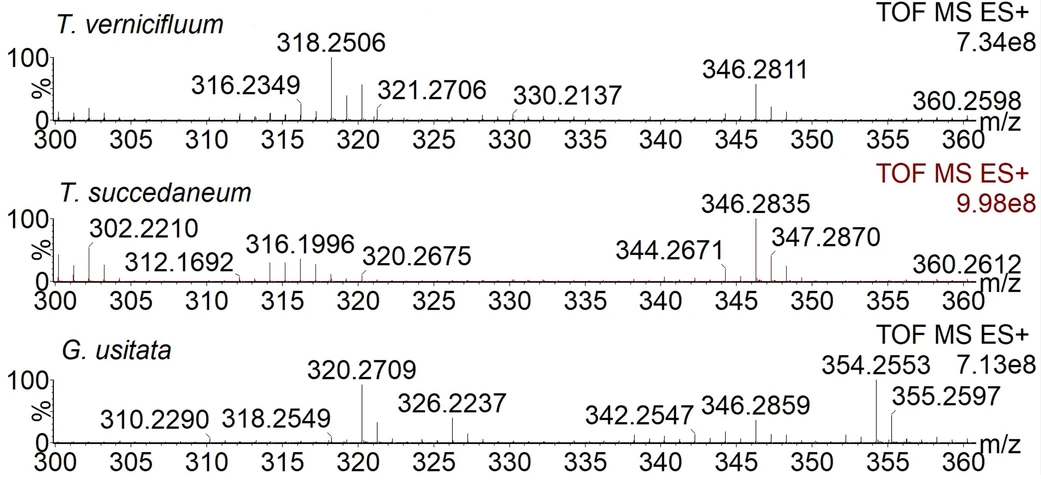 Journal of Cultural Heritage, 77, 2026, 187-196: Fig. 2. Spectra of lacquers prepared from Toxicodendron vernicifluum, Toxicodendron succedaneum and Gluta usitata (samples U, V and B).
Journal of Cultural Heritage, 77, 2026, 187-196: Fig. 2. Spectra of lacquers prepared from Toxicodendron vernicifluum, Toxicodendron succedaneum and Gluta usitata (samples U, V and B).
4.1.4. Study of MS/MS spectra of characteristic markers
For all the standard materials, the most important markers were subjected to fragmentation after the collision induced dissociation (CID) in the Trap collision cell of the mass spectrometer (MS/MS experiments; all fragmentation spectra and observed diagnostic fragments including differences from theoretical masses are given in Supplementary material B).
Signals of fragments with an intensity higher than the specified percentage of well known fragments arising from studied compounds were investigated and identified. Diagnostic fragments are given in Table B.2, Supplementary material B. The threshold signal was set for different compounds. For heptadecyl- and pentadecylcatechols, the threshold signal was defined as 5 % of the signal of dihydroxytropylium that is always an integral component of their collision spectra. For heptadecenyl- and pentadecenylcatechols the threshold signal was 2.5 % of dihydroxytropylium. For pentadecatrienyl catechol it was 100 % of dihydroxytropylium while for dodecylphenylcatechol it was 1 % of dihydroxytropylium. In case of 1-(3-hydroxyphenyl)-12-phenyldodecan-2-one, the threshold signal was defined as 17 % of the signal of hydroxyphenylethan-2-one. Finally, for 10-decylphenylphenol and 12-dodecylphenylphenol, 50 % of the signal of methylphenol was used as the threshold signal. These fragments were used for confirmation of the presence of lacquer compounds in routine measurements (at least one MS/MS spectrum was measured in each sample). The threshold signals were suggested and set in order to reach a sufficient yield of diagnostic fragments for unambiguous identification of individual markers. These signals are intended to be used in long term measurement in practice.
Fragmentation of heptadecylcatechols with 0-3 double bonds (Fig. 3a-d) is similar to that of pentadecylcatechols with same number of double bonds. In all cases, the signals of the characteristic fragments are well separated from the signals of the matrix.
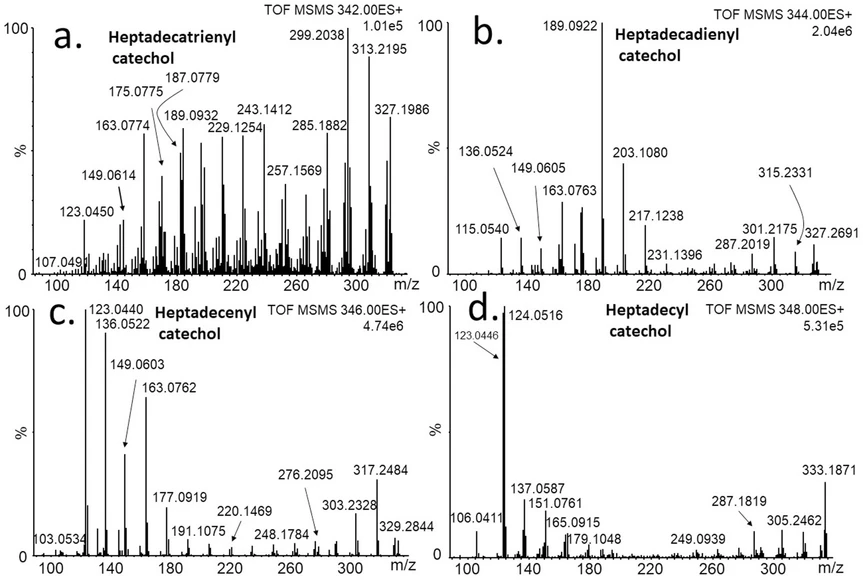 Journal of Cultural Heritage, 77, 2026, 187-196: Fig. 3. Fragmentation spectra of heptadecylcatechols possessing 0-3 double bonds, (MS/MS; m/z of parent ions are: a) 342.2559, b) 344.2715, c) 346.2872, d) 348.3028).
Journal of Cultural Heritage, 77, 2026, 187-196: Fig. 3. Fragmentation spectra of heptadecylcatechols possessing 0-3 double bonds, (MS/MS; m/z of parent ions are: a) 342.2559, b) 344.2715, c) 346.2872, d) 348.3028).
4.2. Principal component analysis
Excellent separation of particular types of Asian lacquers in Score Plot (Fig. 5a-d) was obtained by Principal Component Analysis based on the benzenediol and phenol derivatives described above. The Score Plot contains a set of 20 samples obtained from the Náprstek Museum Prague as well as other samples (BBI, BBO, SBI, SBO, U, V and B) in which lacquer compounds were detected (Table 2). Note that in three other samples from The Náprstek Museum Prague, the lacquer compounds were not found at all.
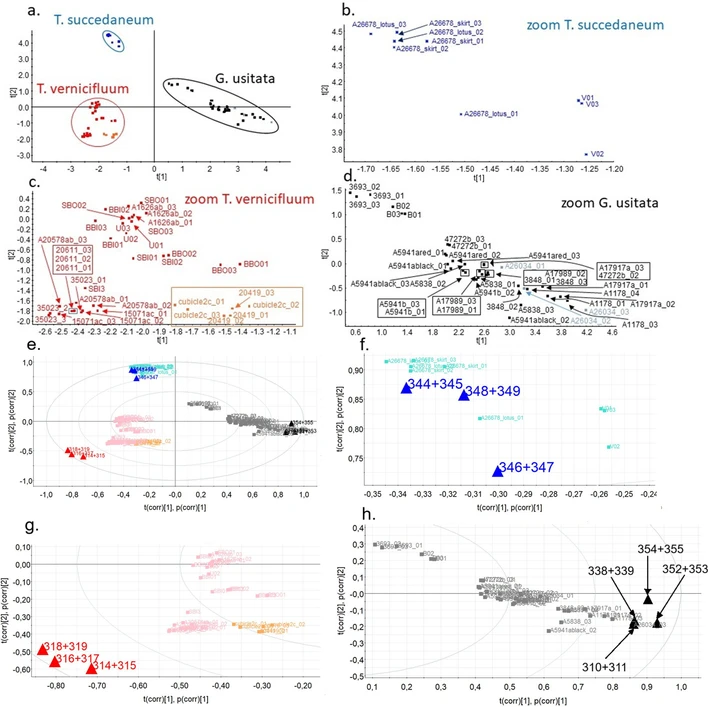 Journal of Cultural Heritage, 77, 2026, 187-196: Fig. 5. Score plot and bi-plot from PCA analysis of lacquers taken from historic objects, a. whole Score PCA plot, b. zoom of the T. succedaneum area, c. zoom of the T. vernicifluum area, d. zoom of the G. usitata area, e. whole PCA bi-plot, f. zoom of the T. succedaneum area in the PCA bi-plot, g. zoom of the T. vernicifluum area in the PCA bi-plot, h. zoom of the G. usitata area in the PCA bi-plot.
Journal of Cultural Heritage, 77, 2026, 187-196: Fig. 5. Score plot and bi-plot from PCA analysis of lacquers taken from historic objects, a. whole Score PCA plot, b. zoom of the T. succedaneum area, c. zoom of the T. vernicifluum area, d. zoom of the G. usitata area, e. whole PCA bi-plot, f. zoom of the T. succedaneum area in the PCA bi-plot, g. zoom of the T. vernicifluum area in the PCA bi-plot, h. zoom of the G. usitata area in the PCA bi-plot.
The significance of suggested markers for the studied lacquers across the whole data matrix was confirmed. The effect of particular markers is reflected in related bi-plots (Fig. 5e-h). Qualitatively, the results are in accordance with the former study obtained by py-GC/MS [1]. Since both cation-radicals and protonated molecules are formed in ASAP source with significant yield of both ion forms, their sum is an input into the data matrix for each substance (e.g., for pentadecenyl catechol, where the input is the sum of intensities I(m/z = 318)+ I(m/z = 319)), see PCA matrix in Supplementary material C. Measured m/z values of the analyzed compounds are in good agreement with the theoretical values calculated for specific elemental compositions (noted differences from the theoretical mass, dtm is in all cases less than or equal to 10 mDa).
5. Conclusion
The combination of atmospheric solids analysis probe with high resolution tandem mass spectrometry proved to be a fast and effective tool for analysis of historical Asian lacquer objects, their resolution and classification. The developed method is based on profiling of phenol and benezenediols derivatives. The fragmentation of alkylphenols and alkylbenzene diols after collision activated dissociation in Trap and Transfer collision cells was described. Scission of alkyl chains and elimination of water represent the main fragmentation processes important for identification. Cyclic ion mobility separation appeared to be a useful tool for “cleaning” of signals of the lacquer ions from the matrix signals allowing their better identification from the fragmentation spectra. Moreover, ASAP-MS and MS/MS allowed detection of diterpenic resins, triterpenic resins and arsenic sulphide pigments present along with Asian lacquers. Principal Component Analysis provided complete separation of samples into three clusters of Asian lacquers in Score Plot. A simple classification approach based on direct comparison of newly suggested classification parameters (CPs) allows reliable differentiation of lacquer samples according to their provenance. The developed method is now implemented in routine controls of lacquered objects and could be applied to the analysis of resins and ambers obtained from archaeological findings.




