Evaluation of a Prototype Orbitrap Astral Zoom Mass Spectrometer for Quantitative Proteomics Beyond Identification Lists
J. Proteome Res. 2025, 24, 11, 5742–5754: Graphical abstract
A prototype Orbitrap Astral Zoom mass spectrometer was evaluated against the standard Orbitrap Astral MS to assess performance beyond conventional identification metrics in quantitative proteomics.
The prototype demonstrated improved ion utilization, sensitivity, and quantitative precision, highlighting its potential as a high-performance platform for data-independent acquisition proteomics and ion-based MS benchmarking.
The original article
Evaluation of a Prototype Orbitrap Astral Zoom Mass Spectrometer for Quantitative Proteomics Beyond Identification Lists
Chris Hsu, Nicholas Shulman, Hamish I. Stewart, Johannes Petzoldt, Anna Pashkova, Deanna L. Plubell, Eduard Denisov, Bernd Hagedorn, Nicolaie E. Damoc, Brendan X. MacLean, Philip M. Remes, Jesse D. Canterbury, Alexander A. Makarov, Christian Hock, Vlad Zabrouskov, Christine C. Wu, Michael J. MacCoss
J. Proteome Res. 2025, 24, 11, 5742–5754
https://doi.org/10.1021/acs.jproteome.5c00593
licensed under CC-BY 4.0
Selected sections from the article follow. Formats and hyperlinks were adapted from the original.
The rapid advancement of mass spectrometry hardware has exposed a critical need to evaluate improvements in new instruments, such as the Thermo Scientific Orbitrap Astral mass spectrometer (MS). (1−6) Frequently, new instruments are evaluated based on the number of identified peptides or proteins using established search tools. (1−13) These metrics, while important, remain rooted in qualitative assessments. Additionally, the search protein and peptide search results will be highly dependent on the tool used and will not necessarily capture differences in instrument performance. (14) Quantitative metrics, such as transmission efficiency, ion beam utilization, and measurement precision or accuracy, are challenging to quantify and compare across instrument platforms. Moreover, new instrument platforms could introduce capabilities that are not immediately compatible with existing data processing workflows; yet, they could offer substantial benefits if analytical tools are adapted to fully utilize them.
To enable direct comparisons between different instrument platforms, we would ideally inject a constant amount of sample into two instruments (or run them under different settings) and then report the number of ions measured by the detector. However, a fundamental challenge lies in the fact that the scale of the reported intensities is often unknown, as they are typically expressed in arbitrary units. While vendors may calibrate detector gain, there remains a need for a universal strategy to convert vendor-specific intensity units into a standardized scale of ions per second (ions/sec). To address this gap, we present a strategy that leverages the relationship between the number of ions measured and the precision of an ion current ratio measurement to derive a correction factor between reported intensity and ions/sec. (15) This calibration can be performed by using a simple infusion experiment on any instrument platform. We use this calibration factor to illustrate both differences and similarities in the reported signal intensities across various Thermo Scientific analyzers and detection technologies.
To capitalize on this calibration of intensity to ions/second, we added new reporting capabilities to the Skyline (16,17) document grid to enable the extraction of peptide-level metrics from liquid chromatography–mass spectrometry data. These metrics include for each peptide (1) the total number of ions in the spectrum at the apex of the chromatographic peak, (2) the number of ions from the extracted precursor > product ion transitions for the target peptide at the chromatographic peak apex, and (3) the number of ions from the target peptide across the chromatographic peak integration boundaries. These metrics can be used to assess both targeted and untargeted measurements.
Using these metrics, we evaluate the effect of modifications to a prototype Orbitrap Astral Zoom mass spectrometer (MS) platform designed to improve the instrument efficiency (Figure 1). This modified instrument was improved in three ways. The overhead time from ion pipelining, which contributes to duty cycle loss, was reduced, resulting in a decrease in cycle time. This enabled the same ion utilization to be performed in less time. Second, the Orbitrap Astral MS was modified to enable preaccumulation of ions in the bent square quadrupole region of the ion source prior to the quadrupole mass analyzer, as described previously. (18) This preaccumulation enables an improvement in the effective ion beam utilization by the instrument. Third, this modified instrument made use of improved signal processing, which could theoretically deconvolute incompletely resolved fragments in the Astral mass spectra. Using HeLa and extracellular vesicle peptide inputs, we benchmarked an Orbitrap Astral Zoom MS prototype across multiple data-independent acquisition (DIA) settings, assessing acquisition rate, ion counts, precursor and protein detection, and quantitative precision and accuracy. Our results demonstrate that the Orbitrap Astral Zoom MS prototype consistently achieves faster cycle times, higher ion sampling, and improved detection performance, highlighting its potential for high-throughput and biologically relevant applications.
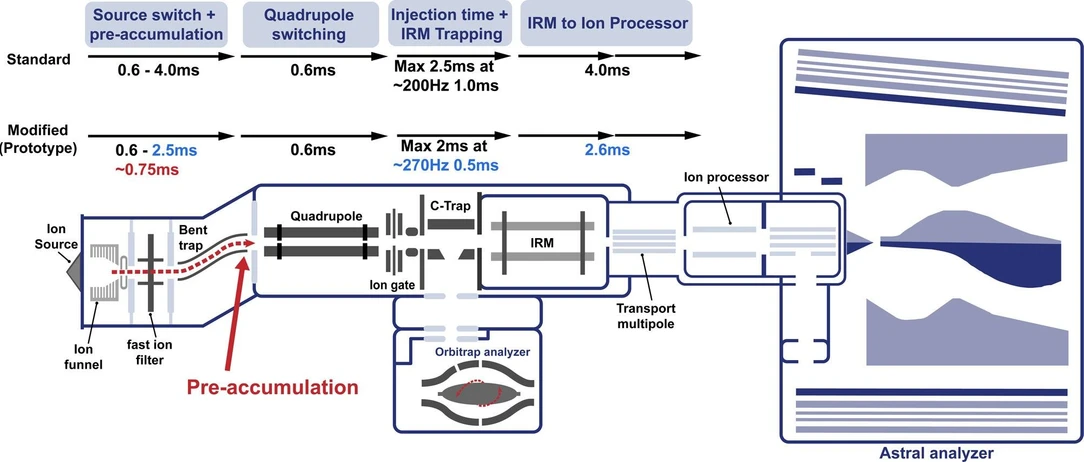 J. Proteome Res. 2025, 24, 11, 5742–5754: Figure 1. Schematic of the Orbitrap Astral Zoom MS prototype. A newly introduced preaccumulation step (in red) allows for longer injection times for the Orbitrap Astral Zoom MS prototype. Additional timing optimizations (in blue) show reduced overhead throughout the ion transfer process compared to the standard Orbitrap Astral MS, enabling higher acquisition rates and improved duty cycle efficiency.
J. Proteome Res. 2025, 24, 11, 5742–5754: Figure 1. Schematic of the Orbitrap Astral Zoom MS prototype. A newly introduced preaccumulation step (in red) allows for longer injection times for the Orbitrap Astral Zoom MS prototype. Additional timing optimizations (in blue) show reduced overhead throughout the ion transfer process compared to the standard Orbitrap Astral MS, enabling higher acquisition rates and improved duty cycle efficiency.
Experimental Methods
Liquid-Chromatography and Mass Spectrometry
All data were acquired on a single Orbitrap Astral Zoom MS prototype instrument coupled to a Thermo Scientific Vanquish Neo UHPLC system. This prototype can be operated in two modes: (1) the standard configuration that replicates the commercially available Orbitrap Astral MS and (2) the prototype configuration with reduced ion pipelining overhead, preaccumulation, and Astral spectral deconvolution. Throughout the manuscript, we refer to these two modes as “Orbitrap Astral MS” and “prototype”. Peptides were separated via a 24 min gradient at 1.3 μL/min, with the following stepwise elution profile: 4–6% Buffer B over 0.7 min, 6–6.5% Buffer B over 0.3 min, 6.5–40% Buffer B over 20 min, 40–55% Buffer B over 0.5 min, and 55–99% Buffer B over 3.5 min for column washing. Following separation, DIA was performed using a cycle comprising a full MS1 acquisition on the Orbitrap detector at 240,000 resolving power, precursor range of 375–985 m/z, 50 ms injection time, and standard AGC. Following each MS1, MS/MS spectra were collected using the Astral detector with the following parameters: precursor mass range of 400–900 m/z, 27% HCD collision energy, and 200% AGC target. For isolation window optimization, three configurations were tested while maintaining consistent cycle times: 2 Th isolation windows with a 4 ms injection time, 3 Th windows with a 6 ms injection time, and 4 Th windows with an 8 ms injection time. The EV dataset was collected using only the 3 Th isolation window with 6 ms injection time setting. The same liquid chromatography and column setup (PepSep C18, 8 cm × 150 μm × 1.5 μm) was used for both the Orbitrap Astral MS and prototype modes. The expected chromatographic peak width was set to 6 s for all sample runs.
Results
Modifications to the Orbitrap Astral MS Improve the MS/MS Acquisition Rate
To assess performance differences between the Orbitrap Astral MS and the Orbitrap Astral Zoom MS prototypes, we compared acquisition rate, acquisition overhead, and cycle time across varying injection times and DIA isolation window configurations. To ensure that the MS/MS acquisitions reached their maximum allowable fill times, we set the H-ESI source to 0 V, disabling electrospray ionization and forcing the system to accumulate ions until the maximum injection time was reached. DIA data were acquired across a range of injection times using two isolation widths: 2 and 4 Th.
Acquisition rate, calculated as the total number of spectra divided by total acquisition time, was consistently higher on the Orbitrap Astral Zoom MS prototype across all conditions (Figure 2A). The largest differences were observed at shorter injection times (1–3 ms). Under the fastest conditions (2 Th with a 4 ms IT), the Orbitrap Astral Zoom MS prototype approached the theoretical acquisition ceiling of 270 Hz, while the Orbitrap Astral MS rate peaked around ∼217 Hz. The Orbitrap Astral MS also showed a larger decline in acquisition rate when using wider isolation windows, likely due to increased acquisition transition costs. To quantify the source of these differences, we calculated acquisition overhead as the difference between MS/MS acquisition duration and the set injection time (Figure 2B). The Orbitrap Astral Zoom MS prototype consistently exhibited ∼1 ms lower overhead compared to the Orbitrap Astral MS across all injection times, and although overhead increased slightly with wider isolation windows, the prototype configuration remained more efficient under all conditions with minimal performance penalty. This improvement in instrument efficiency directly translated into shorter cycle times (Figure 2C), with the Orbitrap Astral Zoom MS prototype consistently achieving faster cycle times across the 400–900 m/z precursor range.
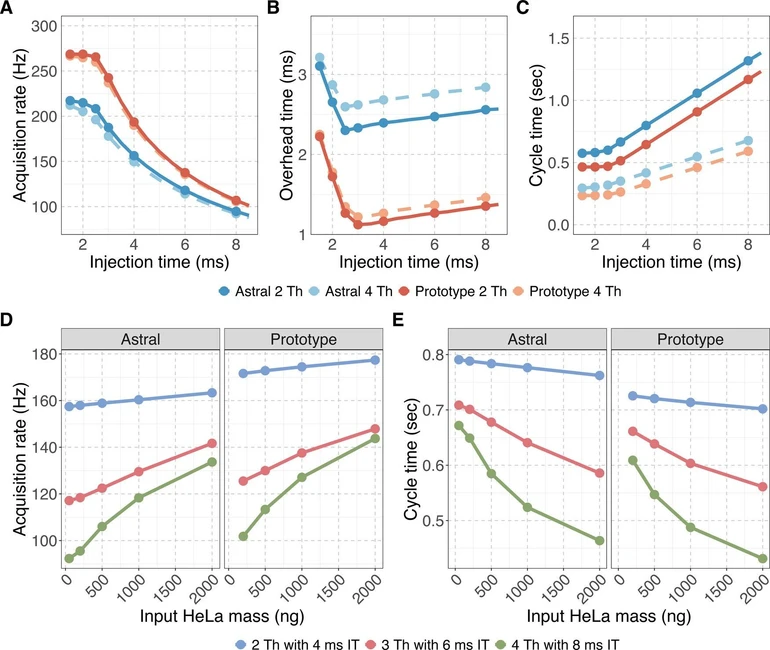 J. Proteome Res. 2025, 24, 11, 5742–5754: Figure 2. The Orbitrap Astral Zoom MS prototype improves MS/MS acquisition rate and reduces acquisition overhead and cycle time. Evaluation of two DIA configurations: narrow (2 Th) and wider (4 Th) isolation windows for (A) MS/MS acquisition rate (Hz), (B) overhead time, calculated as the difference between the mean MS/MS acquisition time and injection time, and (C) cycle time for a 400–900 m/z precursor range. Data in panels A–C represent a single run (one measurement per injection time) with the cycle time values reported as the mean across >30 cycles within that run. Panels (D–E) show the impact of the configuration mode and DIA acquisition scheme on performance across increasing HeLa peptide input for (D) acquisition rate and (E) cycle time are shown for three DIA configurations with set injection times (IT): 2 Th with 4 ms IT (blue), 3 Th with 6 ms IT (red), and 4 Th with 8 ms IT (green). Data in D–E are from a technical triplicate HeLa runs (n = 3).
J. Proteome Res. 2025, 24, 11, 5742–5754: Figure 2. The Orbitrap Astral Zoom MS prototype improves MS/MS acquisition rate and reduces acquisition overhead and cycle time. Evaluation of two DIA configurations: narrow (2 Th) and wider (4 Th) isolation windows for (A) MS/MS acquisition rate (Hz), (B) overhead time, calculated as the difference between the mean MS/MS acquisition time and injection time, and (C) cycle time for a 400–900 m/z precursor range. Data in panels A–C represent a single run (one measurement per injection time) with the cycle time values reported as the mean across >30 cycles within that run. Panels (D–E) show the impact of the configuration mode and DIA acquisition scheme on performance across increasing HeLa peptide input for (D) acquisition rate and (E) cycle time are shown for three DIA configurations with set injection times (IT): 2 Th with 4 ms IT (blue), 3 Th with 6 ms IT (red), and 4 Th with 8 ms IT (green). Data in D–E are from a technical triplicate HeLa runs (n = 3).
Assessing Ion Counts between Orbitrap Astral Zoom MS Prototype and Orbitrap Astral MS
To quantify the number of ions across MS/MS spectra, we implemented a new set of custom metrics in the Skyline document grid that enables ion count calculations for any analyte measured with LC–MS data. As summarized in Table S3, these metrics include LC Peak total ion current (TIC) Area Fragment, Apex Total Ion Count Fragment, LC Peak Total Ion Count Fragment, Apex Analyte Ion Count Fragment, and LC Peak Analyte Ion Count Fragment (also referred to as LC Peak Peptide Ion Count).
For the purposes of this study, we focused on two key metrics: Apex Total Ion Count Fragment and the LC Peak Analyte Ion Count Fragment. The apex value is calculated as the product of the TIC and the injection time for the MS/MS spectrum containing the highest transition intensity. Notably, this value directly mirrors the ion count reported in the raw file at that retention time. The LC Peak Analyte (or Peptide) Ion Count is computed as the sum of the products of transition intensities and injection times across all MS/MS spectra within the peak integration boundaries. This metric approximates the total number of ions contributing to the quantification of a given peptide. Taken together, these metrics establish a quantitative framework for comparing ion counts across instruments, acquisition settings, and peptide intensities. When combined with the appropriate instrument-specific α (alpha) correction factor, they allow for calibrated cross-platform comparisons in units of ions/sec.
The Orbitrap Astral MS instrument uses AGC to regulate the number of ions used in each spectrum. The injection time is adjusted to maintain a target number of ions in each spectrum. To evaluate whether the data collected accurately reflects the AGC target, we examined the apex total ion count per peptide from different amounts of HeLa peptide digests and the three DIA method settings described previously (Figure S6). Both the Orbitrap Astral MS and the Orbitrap Astral Zoom MS prototype were configured with an AGC target of 20,000 ions, and ion counts were calibrated to ions/s using instrument-specific α factors (1.53 for Orbitrap Astral MS and 1.31 for Orbitrap Astral Zoom MS prototype). The median ion counts from both instrument configurations reach the AGC target at 500 ng input, particularly for the 3 Th with 6 ms IT and 4 Th with 8 ms IT settings, suggesting efficient and accurate ion regulation at this input mass. At lower inputs (200 ng), underfilling becomes evident─especially for the narrowest 2 Th window with 4 ms IT─where median ion counts fall below the AGC target as the 4 ms IT is frequently insufficient to reach 20,000 ions. While the 2 Th and 4 ms IT methods might obtain the largest number of protein and peptide detections, they underfill the AGC target most often, except at ≥ 1000 ng loading.
To assess differences in peptide-level ion count, we compared LC Peak Peptide Ion Count Fragment between the Orbitrap Astral MS and Orbitrap Astral Zoom MS prototype across the same data set. Density plots of the mean LC peak ion distributions revealed consistent rightward shifts for the Orbitrap Astral Zoom MS prototype, indicating higher peptide-level ion counts under all conditions (Figure 5A). We extracted the mode of each distribution to quantify the most common ion count under each input mass and DIA setting. Across all input levels and DIA configurations, the Orbitrap Astral Zoom MS prototype yielded higher average mode LC peak ion counts than those of the Orbitrap Astral MS (Figure 5B). The largest gains were observed at lower mass inputs (200–500 ng), where the Orbitrap Astral Zoom MS prototype delivered up to 48% more ions under the 2 Th with a 4 ms IT setting (Figure 5C). Even at higher inputs (≥1000 ng), the prototype maintained at least a 10% increase in the mean modal number of ions per peptide, with the median overall improvement of 23.1%. To further evaluate performance at the individual peptide level, we performed pairwise comparisons of shared peptides, computing the log2 fold-change in the LC peak ion counts between the two instrument configurations. These comparisons showed a consistent positive shift favoring the prototype across all conditions (Figure S7). The greatest increase in log2 ion ratios was observed in the low-to-intermediate ion count range (approximately 10s to 100s of ions), where the prototype likely improves sampling of peptides near the detection limit. In contrast, peptides with high ion abundance (e.g., >1000 ions) already produce strong signals on both instrument configurations, leaving less room for improvement.
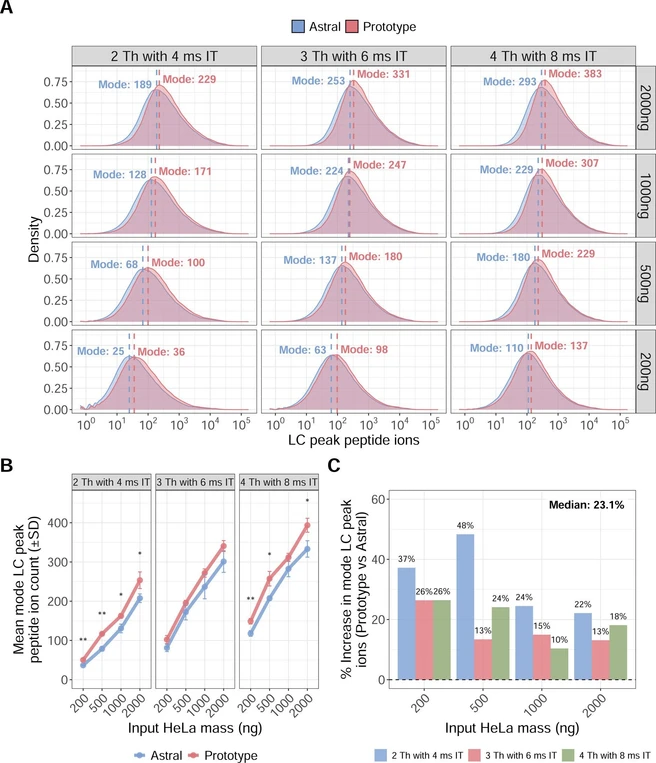 J. Proteome Res. 2025, 24, 11, 5742–5754: Figure 5. The Orbitrap Astral Zoom MS prototype improves LC peak peptide ion counts across sample inputs and DIA methods. (A) Density plots of LC peak peptide ion counts from triplicate runs (n = 3) across various HeLa inputs (200–2000 ng) for each DIA method (2 Th, 3 Th, and 4 Th) comparing the Orbitrap Astral MS and Orbitrap Astral Zoom MS prototype. Vertical dashed lines indicate the mode of each distribution. (B) Line plot showing the mean mode of LC peak peptide ion in triplicate (n = 3) with the standard deviation for the same HeLa peptide inputs and DIA isolation window settings comparing the two configurations. Statistical significance was assessed using unpaired two-sided t tests (*p < 0.05, **p < 0.01). (C) Percent increase in mean mode LC peak peptide ion counts with the reported median percent across all conditions.
J. Proteome Res. 2025, 24, 11, 5742–5754: Figure 5. The Orbitrap Astral Zoom MS prototype improves LC peak peptide ion counts across sample inputs and DIA methods. (A) Density plots of LC peak peptide ion counts from triplicate runs (n = 3) across various HeLa inputs (200–2000 ng) for each DIA method (2 Th, 3 Th, and 4 Th) comparing the Orbitrap Astral MS and Orbitrap Astral Zoom MS prototype. Vertical dashed lines indicate the mode of each distribution. (B) Line plot showing the mean mode of LC peak peptide ion in triplicate (n = 3) with the standard deviation for the same HeLa peptide inputs and DIA isolation window settings comparing the two configurations. Statistical significance was assessed using unpaired two-sided t tests (*p < 0.05, **p < 0.01). (C) Percent increase in mean mode LC peak peptide ion counts with the reported median percent across all conditions.
Discussion
The Orbitrap Astral Zoom MS prototype configuration offers several clear advantages over the standard Orbitrap Astral MS for DIA-based quantitative proteomics. Specifically, the prototype can achieve faster MS/MS acquisition rates, reduced acquisition overhead, and shorter cycle times. Importantly, the enhanced acquisition efficiency also enables a valuable trade-off: users can extend injection times to improve ion sampling while still maintaining acquisition rates comparable to the standard Orbitrap Astral MS. For example, with 2 Th isolation windows, the Orbitrap Astral Zoom MS prototype achieves the same acquisition rate at >7 ms injection time as the Orbitrap Astral MS does at 6 ms. While our study focused on peptide loads ≥200 ng, the improved overhead efficiency and overall instrument flexibility suggest that the Orbitrap Astral Zoom MS is promising for applications that benefit from increased ion efficiency, such as low input or single-cell applications. The strategies described here will be useful for assessing the number of ions that are detected for each peptide and provide a transparent and vendor neutral strategy of assessing sensitivity.
We benchmarked these two instrument configurations by further evaluating the number of precursor and protein detections, the quantitative precision, the ion count, and the quantitative across three different DIA methods and input material. Across all conditions of HeLa peptide input, the Orbitrap Astral Zoom MS prototype detected more precursors and proteins than did the Orbitrap Astral MS with lower median CVs. One of the more striking findings was the ability of the Orbitrap Astral Zoom MS prototype to accumulate more ions per peptide. This was evident across several ion count metrics after calibration by using instrument-specific α correction factors. The Orbitrap Astral Zoom MS prototype not only achieved AGC targets more effectively but also yielded higher total ion counts across the LC peak, suggesting that more ions contributed to each peptide’s quantification. We hypothesize that the increased MS/MS acquisition rate of the prototype improves peptide sampling across the chromatographic peak, thereby contributing to the increased number of ions. These benefits were especially evident in peptides near the detection threshold, where modest gains in ion sampling can significantly improve the detectability and precision. Despite these differences, the Orbitrap Astral MS and Orbitrap Astral Zoom MS prototype modes both provide quantitatively precise measurements.
We demonstrate that mass spectrometers can be calibrated using the well-established relationship between the number of ions and the measurement variance. This calibration standardizes the signal response to ions/sec, enabling meaningful comparisons between different instruments and methods. It also lets us determine how many ions contribute to each measurement, even from instruments that originally had different and arbitrary intensity scales.
It is standard for new quantitative methods to report the CV as a measure of precision. However, these CV values are often calculated from the same small size (frequently fewer than 10 replicates), which limits their reliability. Because of the established relationship between the number of ions and the variance, we propose that reporting the number of ions used in each measurement provides a valuable complement beyond standard precision figures of merit. When both the ion count and precision are reported together, researchers can better assess whether a reported CV might be misleadingly optimistic due to small or biased sampling. This dual reporting approach offers a more complete picture of the measurement quality and reliability.
Additionally, both instrument configurations demonstrate strong quantitative accuracy across a dynamic range of peptide inputs and sample types, including matrix-matched human EV samples. While the Orbitrap Astral Zoom MS prototype outperforms the Orbitrap Astral MS in most metrics that we tested, the Orbitrap Astral MS remains a robust and reliable platform for DIA-based proteomics.