Oligos Made Easy - Part 7: Large Scale AEX

KNAUER: Oligos Made Easy - Part 7: Large Scale AEX
Why Anion Exchange Chromatography Is a Strong Choice for Large-Scale Oligonucleotide Purification
Oligonucleotide therapeutics are becoming increasingly important in modern drug development. Short DNA- and RNA-based medicines are moving through clinical pipelines in growing numbers, and several have already received FDA approval. As this field expands, one question becomes increasingly important: how can these molecules be purified efficiently, reproducibly, and on a manufacturing scale?
One of the most reliable answers is anion exchange chromatography (AEX).
In earlier discussions, we looked at the fundamentals of oligonucleotide analysis and the core principles behind AEX separations. Here, the focus shifts to preparative purification at larger scale and to the practical issues that arise when moving from laboratory development toward production. As an example, this article highlights the purification of an oligo(dT) sample on the liter scale, demonstrating how AEX can deliver both high purity and strong recovery.
Why purification is so important
Following chemical synthesis, crude oligonucleotide material contains a mixture of desired product, truncated sequences, and other synthesis-related impurities. For pharmaceutical use, especially in clinical and regulated settings, this crude material must be refined to a much higher purity level.
Although multiple HPLC approaches are available for oligonucleotide purification, not every method is equally suitable for scale-up. Some techniques work well in analytical or small-scale formats but become less practical when transferred to larger production environments. What is needed is a purification strategy that combines high performance with straightforward scalability.
What makes AEX attractive?
Anion exchange chromatography has established itself as an effective purification method for oligonucleotides, especially when larger sample volumes are involved. Its main strengths include:
- High loading capacity, allowing large sample amounts to be processed efficiently
- Strong separation performance, enabling high-purity product recovery
- Robust scalability, from development work through to GMP-oriented production
- More environmentally friendly operation, since aqueous buffer systems can be used instead of harsher organic solvents
These features make AEX a practical option for laboratories and manufacturers seeking a reproducible purification platform that can grow with the needs of a project.
Example: large-scale purification of oligo(dT)
To illustrate the approach, an oligo(dT) sample was purified on the liter scale. Oligo(dT) is widely used in molecular biology, for example in mRNA isolation and as a primer in cDNA synthesis.
Before chromatography, the sample required the usual preparation steps: cleavage from the solid support, deprotection with ammonia, and subsequent neutralization. The purification itself was performed on the AZURA® Prep HPLC system, which is designed for high-throughput preparative workflows and offers precise gradient formation, UV detection, and automated fraction collection.
 KNAUER: AZURA preparative HPLC system
KNAUER: AZURA preparative HPLC system
The separation used a strong anion exchange resin with gradient elution based on a phosphate buffer and sodium bromide as the counterion. Sodium bromide was selected because it is less aggressive toward stainless steel components than chloride, helping protect instrument hardware during repeated operation. For optimal resolution, anion exchange particles in the 10–15 µm range are typically preferred.
Performance in practice
The results clearly demonstrate the suitability of AEX for oligonucleotide purification at scale.
- Purity before purification: 89%
- Purity after AEX purification: 95%
- Yield after purification: 90%
These values show that the method can effectively improve product quality while maintaining a high recovery, which is a key requirement in preparative and manufacturing settings.
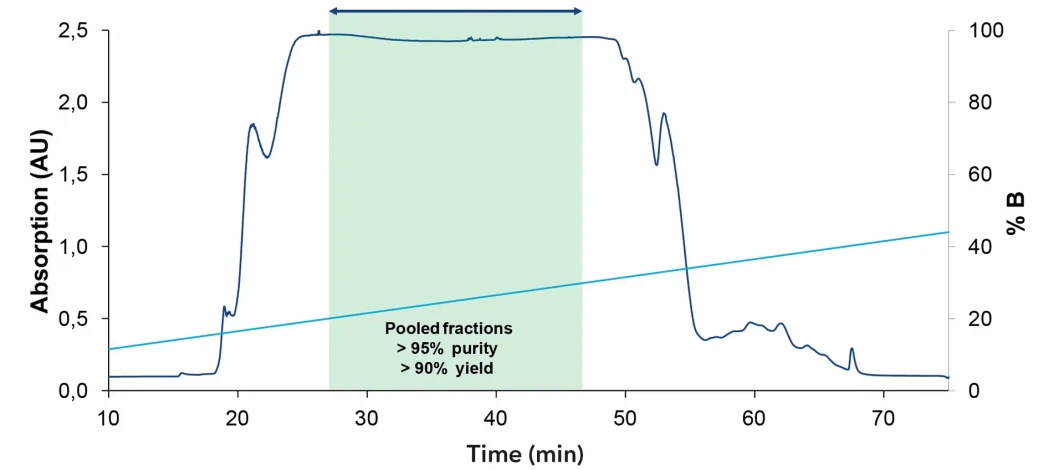 KNAUER: Chromatogram of oligonucleotide purification from 1 l sample
KNAUER: Chromatogram of oligonucleotide purification from 1 l sample
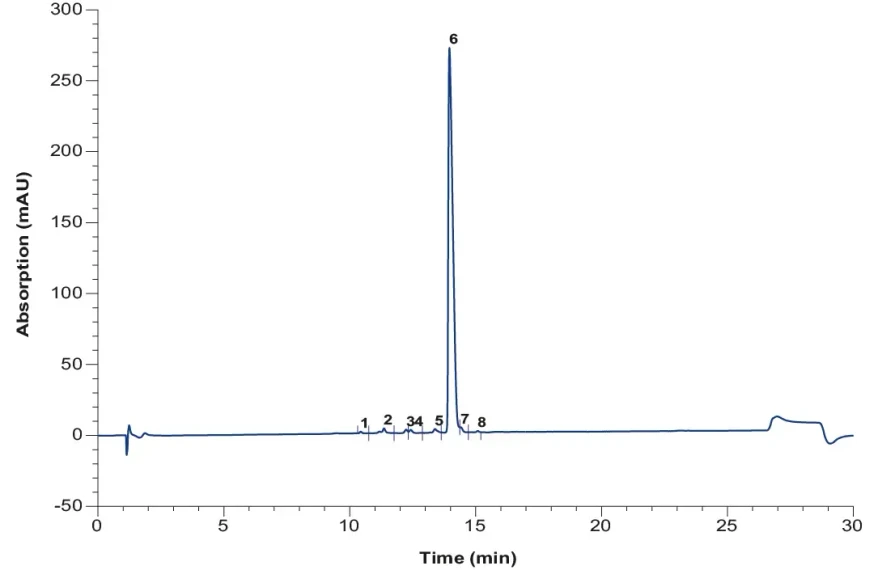 KNAUER: Analytical chromatogram of purified and pooled oligo (dT) after AEX.
KNAUER: Analytical chromatogram of purified and pooled oligo (dT) after AEX.
Important limitations to keep in mind
Like any chromatographic method, AEX also has its limitations and process considerations.
- Phosphodiester vs. phosphorothioate separations can be challenging, and resolution may decrease for longer or more heavily modified sequences.
- Aggregation effects caused by secondary structures can reduce purification efficiency.
- Because AEX is typically performed under aqueous conditions, there is also an increased need to monitor and control endotoxin-related process risks.
These factors do not diminish the value of AEX, but they do underline the importance of careful method development and process control.
Final thoughts
For oligonucleotide-based therapeutics, purification is not just a supporting step — it is a critical part of the path from crude synthesis to clinical or commercial material. The purification strategy must be scalable, robust, and capable of consistently delivering high-quality product.
Anion exchange chromatography meets many of these demands. It offers a compelling balance of purity, yield, scalability, and operational practicality, making it an important tool for both development and production environments.
For teams preparing material for research, clinical studies, or GMP manufacturing, AEX on AZURA® Prep systems represents a strong and practical route toward reliable oligonucleotide purification.
For additional details, the related application note “Oligonucleotide Purification via Ion Exchange” provides more information on the method.
- For further information on this topic, please contact the author at [email protected]
- If you are planning to build or optimize an oligonucleotide workflow, you can also contact [email protected]




