Menu
More information
WebinarsAbout usContact usTerms of use
LabRulez s.r.o. All rights reserved. Content available under a CC BY-SA 4.0 Attribution-ShareAlike
Inactive

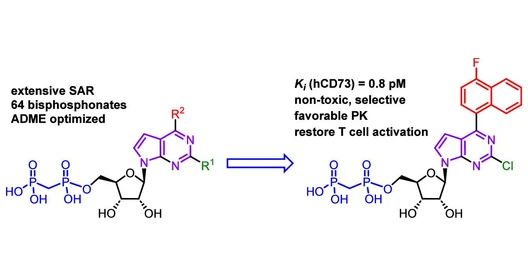
Shimadzu MALDI-Based Solutions for Food Quality Control
We will present some examples of MALDI-based applications for food quality control aiming at tackling issues of food adulteration and safety, using a small benchtop MALDI-TOF instrument from Shimadzu.
RECORD
|
Already taken place Th, 11.4.2024
Shimadzu Corporation


Empower Tips: Ask Me Anything About Using and Reviewing Information in Empower Software Audit Trails
In this webinar, you will gain an understanding of how to use the information captured in the various Audit Trails within Empower Software and in the Empower Message Center.
RECORD
|
Already taken place Th, 11.4.2024
Waters Corporation


Score easy chemistry automation driven by streamlined software with Junior & Big Kahuna
Learn how Big Kahuna and Junior, powered by LEA software, simplify lab automation. Discover flexible workflows, scripting, and AI-ready integration to streamline chemistry processes.
RECORD
|
Already taken place We, 10.4.2024
Unchained Labs

Related content
More information
WebinarsAbout usContact usTerms of use
LabRulez s.r.o. All rights reserved. Content available under a CC BY-SA 4.0 Attribution-ShareAlike